
Ever stop to think about the unsung heroes powering our lives? From the tiny button cells in your remote to the mighty slabs under the hood of your car, batteries are everywhere. They're like magical little boxes that just work. But have you ever poked around a bit and wondered, "Hey, what exactly is inside these things? And wait, isn't there, like, acid in there?"
You're not wrong! For many common types of batteries, especially the powerful ones, acid plays a pretty crucial role. But let's unwrap this mystery without getting too bogged down in super-heavy chemistry terms. Think of this as a relaxed chat about the fizzy, zappy secrets of our portable power sources.
The Big Player: Sulfuric Acid
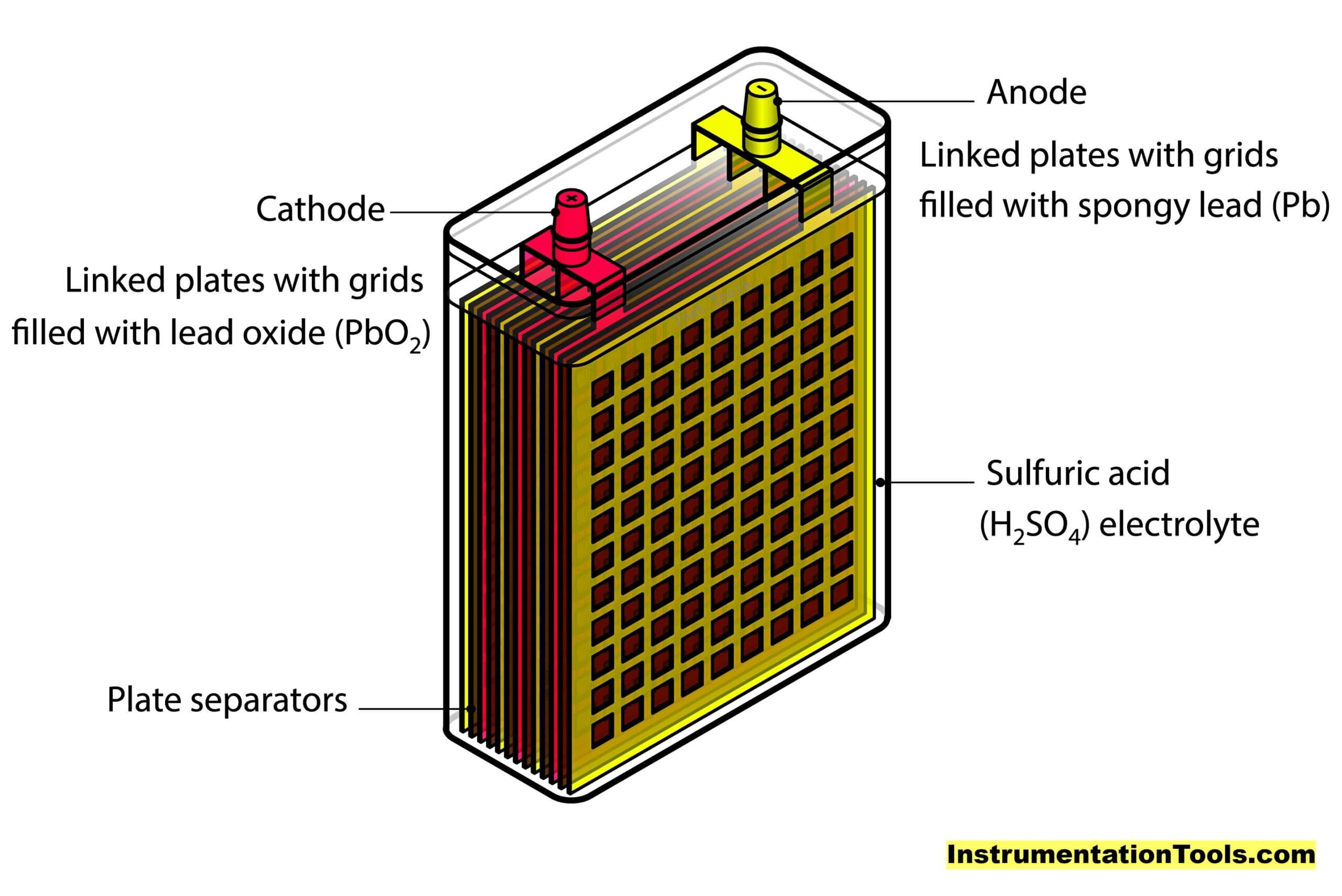
When most people think of "battery acid," they're probably picturing the stuff inside a traditional car battery. And for good reason! The workhorse behind those powerful jolts that get your engine roaring is typically sulfuric acid.
Yup, the same stuff you might have vaguely remembered from a high school science class. In a lead-acid battery (like the one in your car), this sulfuric acid isn't just chilling out. It's actually mixed with water, forming an electrolyte solution. This solution is what allows the magic to happen, acting as a superhighway for charged particles (ions) to zip between the lead plates inside the battery.
Imagine it like a tiny, bustling city where the sulfuric acid is the crucial road network. Without it, the "cars" (electrons) can't get where they need to go, and your battery just sits there, feeling lonely and powerless. Pretty cool, right? It’s not just an inert liquid; it’s an active participant in the chemical dance that generates electricity.

Not All Acids Are Created Equal (And Some Aren't Even Acids!)
Now, here’s where it gets a little more nuanced and, frankly, even more interesting. While sulfuric acid is king in the automotive world, it's important to remember that not every single battery contains sulfuric acid. In fact, many popular batteries don't contain liquid acid at all!
Take your smartphone, for example, or your laptop. These rely on lithium-ion batteries. Do they have a sloshing liquid acid inside? Nope! These modern marvels typically use an organic solvent containing a lithium salt (which isn't an acid in the traditional sense) as their electrolyte. It’s more like a highly specialized, conductive goo or gel that allows lithium ions to travel back and forth, charging and discharging the battery. So, no "acid" in the sulfuric acid sense here!
And what about the everyday AA or AAA batteries you use for remotes and flashlights? Those are usually alkaline batteries. The electrolyte in these is typically potassium hydroxide. Here’s the fun twist: potassium hydroxide isn't an acid at all! It's actually a base, which is the chemical opposite of an acid. Mind blown, right? So, while we often generalize and say "battery acid," it's a bit of a misnomer for many of the batteries we interact with daily.
Why the "Acid" (or Base) Matters: The Flow of Power
So, why bother with these special chemical solutions – be they acids, bases, or specialized salts – in the first place? It all comes down to the fundamental job of a battery: generating an electrical current.

Batteries work through a process called electrochemistry. In simple terms, they facilitate a chemical reaction that involves the movement of electrons from one place to another. The electrolyte (our acid, base, or salt solution) is the vital medium that allows this movement to happen internally, completing the circuit within the battery itself.
Think of it like this: the solid parts of the battery (the electrodes) are like two separate islands. The acid or electrolyte is the ocean in between. Without that ocean, the tiny "ships" (ions) carrying the electrical charge can't travel between the islands to complete their journey and power your device. It’s a beautifully orchestrated chemical dance that turns potential energy into the kinetic energy of flowing electrons.
Respect the Power, Understand the Wonder
Of course, whether it's sulfuric acid, potassium hydroxide, or a lithium salt solution, the chemicals inside batteries are powerful. That's why we always treat batteries with respect and handle them properly. They're designed to store and release significant amounts of energy!
But understanding what's inside takes them from being mysterious black boxes to fascinating mini-power plants. It's not just "acid"; it's a precisely engineered chemical environment that allows electrons to do their amazing work, bringing light to our homes, starting our cars, and keeping us connected.
So next time you pick up a battery, give a little nod to the clever chemistry happening within. Whether it's the classic sulfuric acid or a more modern electrolyte, it's all part of the incredible science that makes our modern world run! Isn't that just awesome?