
Ever heard the word polymer? It might sound like something straight out of a science fiction movie, a secret code whispered by brilliant scientists in lab coats. But trust us, polymers are not only real, they are absolutely everywhere around you! They are the unsung heroes of our modern world, quietly making your life better and way more fun.
So, what exactly is a polymer? Let's break down this magnificent word, because understanding its name is like unlocking a tiny superpower. The word polymer comes from ancient Greek, and it’s surprisingly straightforward when you look closely. It’s like a secret handshake with science!
Think of the first part: "Poly-". You've heard it before, right? Like in polygon (many sides) or polyglot (many languages). So, "poly" simply means many. It's like a crowd of tiny, important things!
Then there's the second part: "-Mer". This little gem comes from the Greek word meros, which means parts or units. So, if we put it all together, what do we get?
Polymer literally means "many parts."
How cool is that? It's just a bunch of little parts, joined together in a spectacular fashion! It's less intimidating than it sounds, and way more exciting.
Now, what are these "parts"? Each individual "part" or "unit" is called a monomer. Again, "mono" means one. So, a monomer is just a single building block, a lone ranger of the molecular world. Imagine a single LEGO brick just sitting there, waiting for its destiny.
When these single monomers decide to get together and party, they link up in long, magnificent chains. It's like a molecular dance party where everyone holds hands! These giant, interconnected chains are what we proudly call a polymer.
Imagine this: Think about a string of beads. Each individual bead? That's a monomer, small and ready for action. The entire glorious necklace, sparkling and complete? That's a polymer! Or picture a super long train. Each individual train car is a monomer, and the whole magnificent train chugging along is the polymer. See? Simple!

It's the ultimate molecular linking game! These tiny individual units link up, one after another, creating wonderfully long, often repeating structures. They can be straight lines, branched trees, or even intricate networks, all from humble beginnings.
The real magic of polymers lies in this "many parts" structure. By linking simple, small molecules together, chemists can create materials with incredible properties. They can be super strong, incredibly flexible, wonderfully lightweight, or even delightfully squishy. It’s like having a molecular toolkit that can build almost anything you can dream up!
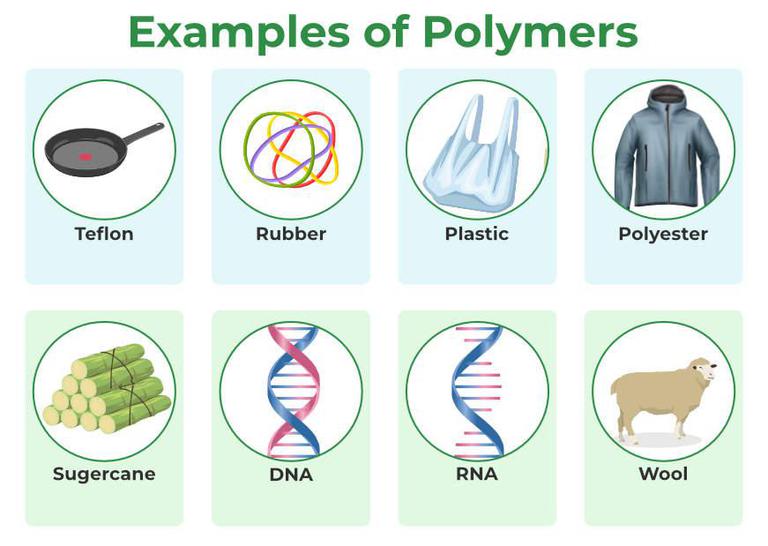
Let's talk about some of our favorite polymer pals. You might be surprised just how many familiar faces pop up once you know their secret identity. They’ve been silently shaping your world, making it comfier, safer, and definitely more fun every single day.
Natural Polymers: Nature's Masterpieces
Did you know that polymers aren't just something cooked up in a lab? Oh no, Mother Nature was the original polymer queen! She's been creating these incredible "many-part" structures for billions of years. She practically invented molecular construction!
The Building Blocks of Life Itself!
Take, for example, DNA. Yes, that incredible double helix that holds all your genetic information? It's a polymer! Each tiny nucleotide (adenine, guanine, cytosine, thymine) is a monomer. When they link up, you get the amazing, life-giving polymer we call DNA. How wild is it that you are essentially a walking, talking polymer marvel?
Think about it: every cell in your body is packed with these complex, informational polymers. They dictate everything from your eye color to your ability to wiggle your toes! Talk about a vital role for "many parts" joining forces.
The Power of Proteins
What about proteins? Your muscles, your hair, the enzymes that help you digest food – they're all made of proteins. Each protein is a magnificent polymer, built from smaller units called amino acids. These amino acids are the monomers that link up to form the complex, folded structures of proteins. They're like tiny, molecular construction workers building your body, cell by cell!
These protein polymers are incredibly diverse, performing a dazzling array of tasks in your body. From transporting oxygen in your blood to giving your skin elasticity, they are truly the multitaskers of the molecular world. They are little powerhouses made of many parts!
Plant-Based Powerhouses
Ever wondered what makes wood so strong, or cotton so soft? That's right, cellulose! Cellulose is a giant polymer made from repeating units of a sugar molecule called glucose. It's what gives plants their structure and strength, making them stand tall and proud.
Imagine tiny sugar molecules linking hands over and over again to build something as mighty as an oak tree or as comfy as your favorite cotton t-shirt. It’s pure natural magic!
And don't forget starch! That energy storage in potatoes, rice, and grains? Also a polymer of glucose, just linked a little differently. So, when you're enjoying a tasty carbohydrate, you're actually fueling up on natural polymers, ready to give you energy!
Synthetic Polymers: Human Ingenuity Unleashed
While nature gave us a head start, humans quickly learned how to get in on the polymer action. Scientists and engineers, inspired by nature's cleverness, started designing and creating their own "many-part" materials in labs. And oh boy, did they unleash a revolution that changed everything!
The Plastic Fantastic!
When most people think of polymers, they often think of plastics. And for good reason! Plastics are perhaps the most famous group of synthetic polymers, and they've completely transformed our world. From the bottle you drink water from to the dashboard of your car, plastics are everywhere, making life easier and more convenient.
Think of polyethylene (the "PE" in your plastic bags and milk jugs), polypropylene (yogurt containers, car parts), or PET (water bottles, clothing fibers). Each of these is a unique polymer, made by linking different monomers in specific ways. They are like cousins in the big polymer family, each with its own special talents.
It's like having an infinite set of LEGOs, but instead of just bricks, you have different shapes and sizes of connecting pieces, allowing for endless possibilities in what you can build and create!
These synthetic polymers can be incredibly lightweight, waterproof, shatterproof, and super durable. They’ve allowed us to create everything from protective gear for athletes to the incredibly light materials used in spacecraft. They are truly versatile!
Beyond the Bottle: Other Amazing Synthetics
But synthetic polymers are so much more than just bottles and containers. Think about nylon! It's a synthetic polymer that's super strong and durable, perfect for ropes, clothing, and even the bristles on your toothbrush. It’s tough stuff, built to last.

Or how about silicone? It's known for its incredible flexibility, heat resistance, and waterproof properties. This makes it perfect for bakeware, stretchy sealants around your windows, and even life-saving medical devices. It’s the ultimate bend-and-don’t-break polymer!
Even the super-sticky glue that holds your sneakers together or the vibrant paint on your walls likely contains synthetic polymers. They are the hidden heroes, providing structure, protection, and performance in countless products you interact with every single day. They are truly all around us!
The Ever-Shifting, Ever-Useful World of Polymers
So, the next time you hear the word polymer, don't feel intimidated. Feel enlightened! Remember, it just means "many parts," tiny units linked together to create something much bigger and much more incredible. It’s a simple concept with monumental impact.
They are the ultimate molecular shapeshifters, capable of being soft or hard, stretchy or rigid, transparent or opaque. Their versatility is truly astonishing, making them essential for almost every aspect of modern life. Without them, our world would look incredibly different!
From the DNA in your cells that makes you uniquely you, to the plastic keyboard you might be typing on right now, polymers are working tirelessly behind the scenes. They truly are the molecular superstars, making our world safer, more convenient, and undeniably more interesting. So go ahead, give a little nod of appreciation to the amazing world of polymers – they certainly deserve it!