
The Secret Life of Things You Hold
Ever pick up something and just know it's got some heft to it? You know, the kind of weight that makes you go, "Whoa, this isn't messing around."
That undeniable feeling often comes from something called density. And today, we're shining a playful spotlight on a particularly weighty character: copper, a true champion of "having substance."
Copper: More Than Just a Pretty Face
Think about a shiny new penny – back when pennies were still mostly copper, of course. Or perhaps those trendy Moscow Mule mugs clinking cheerfully in your kitchen.
Then there are the less glamorous but equally vital roles: the copper pipes diligently hiding in your walls, carrying water and doing their heavy-duty, unseen work every single day.
It’s everywhere, isn't it? And every time you interact with it, there's this underlying sense of... substance. A solidness you can almost feel in your very bones.
It’s not flimsy, not delicate; it feels undeniably purposeful, thoroughly dependable. Almost like a trusty old friend who always shows up with exactly the right amount of gravitas.
Density Demystified (Kind Of)
Now, "density" sounds like something you'd learn in a very serious science class. You know, with goggles, perhaps a bubbling beaker, and a stern-faced professor making important declarations.
But really, it's just a wonderfully fancy way of saying how much "stuff" is squished into a certain amount of space. Think of it as a competitive packing efficiency contest.
Imagine holding a fluffy feather in one hand and a tiny, unassuming pebble in the other. Which one feels significantly heavier? The pebble, obviously!
Even though the feather might take up more space, the pebble has far more actual mass packed into its smaller volume. That, my friends, is density in action, plain and simple.

It's not about how big something looks, but rather how much "oomph" it genuinely packs per cubic inch. It's all about how tightly those particles are crammed together.
Why Copper Feels So... Solid
So, where does our friend copper fit into this impressive packing efficiency show? Turns out, copper atoms are a bit like extremely polite, well-organized partygoers.
They don't spread out much, hogging all the available space. Instead, they prefer to huddle together, close-knit, making the overall material very compact and delightfully substantial.
There’s not a lot of wasted, empty space between these metallic little socialites. They're all about being cozy, efficient, and taking up as little room as possible while still being present.
This atomic snuggle-fest is precisely why copper feels so remarkably substantial when you pick it up. It’s got a lot of "you" (meaning, mass) in a surprisingly small package.
This inherent compactness is exactly what gives copper its distinctive, satisfying heft, whether it's a pipe, a wire, a piece of intricate jewelry, or a grand piece of art.
The Big Reveal: The Actual Number! (Or Is It?)
Okay, okay, you’ve waited patiently. You’ve endured my analogies about feathers and pebbles. You are now ready for the cold, hard numbers for copper's density.
And here it is, dear reader, drumroll please, a number that scientists have meticulously measured and agreed upon after countless experiments and precise calculations:

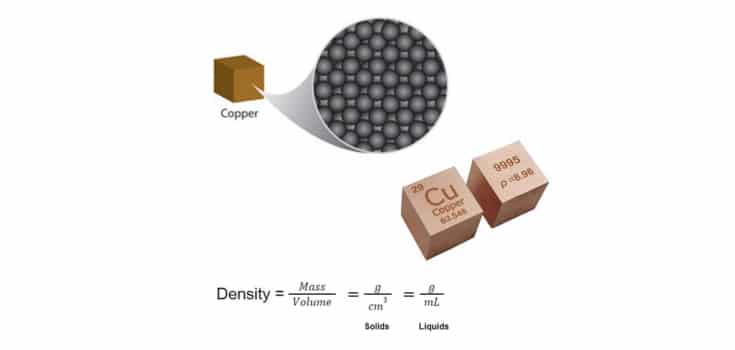
Pure copper's density is approximately 8.96 grams per cubic centimeter.
Yes, 8.96 g/cm³. A beautiful, precise number! A triumph of scientific measurement! A testament to human curiosity and meticulous observation!
For those who absolutely adore precision and numerical accuracy, that number is indeed a masterpiece. It's definitive, it’s wonderfully quantifiable, and it's a scientific fact you can certainly take to the bank.
But here's my controversial, perhaps even wildly unpopular, opinion, which I sincerely hope you'll secretly agree with: Does knowing that exact number, 8.96, truly change your life in any meaningful, earth-shattering way?
For most of us, probably not. Unless, of course, you're an engineer designing a spacecraft where every single gram counts, or a jeweler meticulously weighing precious metals for a bespoke creation.
The Unpopular Truth: The Feeling Trumps The Figure
I propose that the feeling of copper's density is far more relevant to our daily existence than conscientiously remembering the specific digits 8.96.
When you pick up a hefty copper pot from the kitchen cupboard, do you honestly think, "Ah, precisely 8.96 grams per cubic centimeter, how wonderfully dense!"?
Or do you think something more along the lines of, "Wow, this pot means serious business! I could probably use this to fend off a small, very confused, yet surprisingly persistent dragon!"?

I’m betting on the dragon-fending thought, aren't you? Because that's what density truly communicates to us: the intrinsic weight, the undeniable solidity, the sheer substance of an object.
It's an intuitive understanding, a primal appreciation for how much "there" is to something, regardless of its precise measurements or formal definition.
This isn't about shunning science, mind you. It's about prioritizing the incredibly valuable human experience of science over the sometimes-unnecessary rote memorization of obscure figures.
Why We Love That Heavy Feeling
There's a certain deeply satisfying psychological response to holding something dense. It just feels premium. It feels durable. It feels like it's built to last for generations, a true heirloom.
That's the silent magic of copper's high density at play. It whispers promises of quality, permanence, and an enduring strength that simply can't be faked.
It's why heavy, well-balanced cutlery feels infinitely nicer to use than flimsy plastic. It elevates the simple, everyday act of eating into something more substantial and enjoyable.
It’s why a solid wood door feels so much more secure and reassuring than a hollow, lightweight one. You can physically feel the protection it offers, the barrier it creates.
Copper, with its impressive density, speaks this universal language fluently, making us feel more connected to the objects we interact with daily in a tangible way.
It hints at meticulous craftsmanship, at materials chosen specifically for their inherent, superior qualities, not just for their cheapness or the ease of their production.
Embrace the Density, Forget the Digits (Mostly)
So, the next time you encounter a piece of copper – perhaps a beautiful, gleaming statue, an ancient coin, or even a humble electrical wire – don't stress about recalling its exact density.
Instead, just appreciate the feeling. The satisfying, reassuring weight in your hand. The undeniable solidity. That, my friends, is the real, deeply personal takeaway here.
It’s a wonderful reminder that some things in life are just wonderfully, gloriously dense. And that's not just a scientific fact; it's a profoundly tactile and enjoyable pleasure.
Let's all collectively agree: the experience of copper's density is far more entertaining and genuinely relevant to our everyday lives than memorizing its precise numerical value.
It's our little secret now, a shared understanding among those of us who truly appreciate the good, honest heft of things. And now you know it too, so spread the word (or don't, it's a secret after all!).
Go forth and appreciate the beautifully weighty world around you! You're welcome for this enlightenment. Or maybe I'm welcome for giving you explicit permission to conveniently forget a specific number.
Either way, I think we can all agree, that's a delightfully win-win situation. Now go pick up something copper and just feel it – you won't regret it!