Ever found yourself stirring a cup of tea with a spoon, or perhaps admiring an antique pewter goblet, and wondered about the very stuff they’re made of? It might sound a bit nerdy, but diving into the world of elements and their properties, like the atomic mass of tin, can actually be quite a fascinating journey! It’s like peeking behind the curtain of the universe to understand the fundamental building blocks of everything around us. Far from being a dry scientific fact, understanding something like tin's atomic mass helps us appreciate why this versatile metal behaves the way it does in our everyday lives, from the solder connecting electronics to the bronze in a beautiful sculpture.
So, who benefits from knowing about tin's atomic mass? For beginners, it's a super accessible way to start understanding the periodic table and what those numbers actually mean. It demystifies chemistry, showing that even complex-sounding terms are just ways to describe the real world. For families, it can spark curiosity in kids – "Hey, did you know that the metal in these 'tin' cans (which are mostly steel, by the way, but coated with actual tin!) has a specific weight to its tiniest particles?" It's a great conversation starter about how everything is made of atoms. And for various hobbyists – think budding jewelers working with pewter, electronics enthusiasts soldering circuits, or even amateur historians studying ancient artifacts – knowing the atomic mass helps them understand the material's density, how it alloys with other metals, and its overall physical properties. It's the kind of foundational knowledge that makes other aspects of their hobbies click into place.
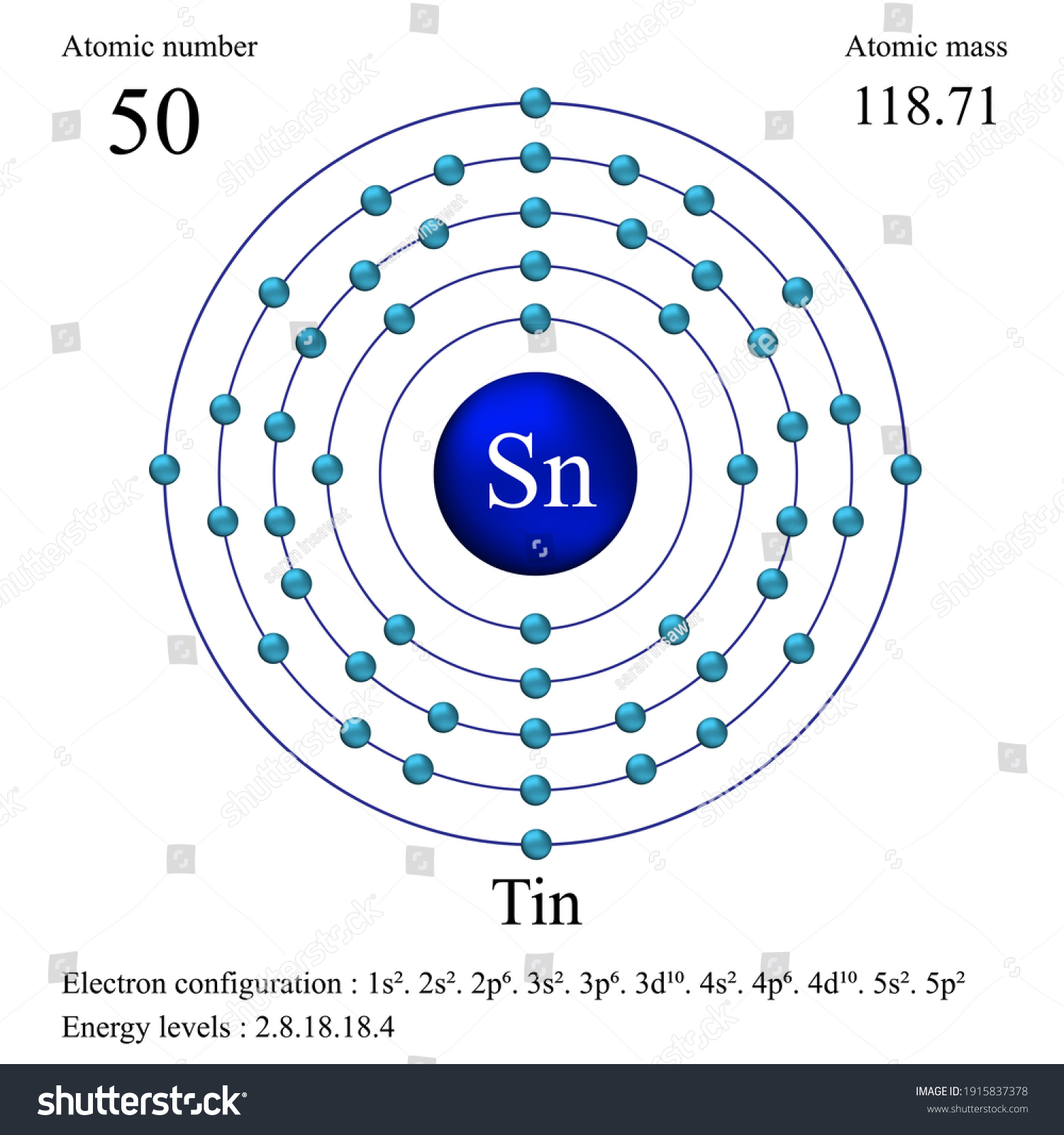
Let's get to the nitty-gritty: the atomic mass of tin is approximately 118.71 atomic mass units (amu). Now, why isn't it a nice, round whole number? This is where tin gets really interesting! Unlike some elements, tin has an impressive ten stable isotopes – that means there are ten naturally occurring versions of tin atoms, each with a slightly different number of neutrons (and thus a different mass). The 118.71 amu you see on the periodic table is actually a weighted average of all these isotopes, reflecting how abundant each one is in nature. This is a brilliant example of how science accounts for the nuances of the natural world. To put it in perspective, tin is significantly heavier than iron (around 55.84 amu) but lighter than lead (around 207.2 amu), which explains why it feels relatively dense but not as 'heavy' as lead in a similar volume. This weighted average influences everything from tin's melting point to how it reacts in different chemical processes, making it ideal for many uses, like protecting steel from corrosion or forming low-melting-point alloys.
Ready to start your own mini-exploration? Here are a few simple, practical tips. First, grab a periodic table (you can easily find one online!) and locate tin, element symbol Sn. Notice its atomic number (50, the number of protons) and its atomic mass. Then, compare it to neighboring elements or elements you interact with daily, like copper or iron, to get a sense of scale. A fun exercise is to look up some of tin's many isotopes and see how they contribute to that average number. Finally, next time you see something made with tin – perhaps a can of soup, some solder, or a piece of pewter jewelry – take a moment to appreciate the tiny, invisible atoms, each contributing its 118.71 amu to the object you're holding. It's a little mind-bender!
Understanding something as specific as the atomic mass of tin might seem like a small detail, but it opens up a huge window into how our world is constructed. It’s a wonderful reminder that even the simplest objects have complex, fascinating stories at their atomic heart. So, go ahead, enjoy the simple pleasure of looking at everyday materials with new, more informed eyes!