
Okay, picture this: You’re on a roll, feeling all Bob the Builder, trying to hang that new shelf your significant other (or just your inner perfectionist) has been eyeing. You grab what you think is a sturdy drill bit, maybe one of those cheap ones from a bargain bin, and go to town. Zzzzzzzt! A puff of smoke, a whiny groan from the drill, and your bit is twisted into a pathetic little pretzel. Not ideal, right?
Then, you dig around, find an old, trusty drill bit – probably a dull grey one, maybe a little rusty, but feels substantial. You slot it in, try again. Rrrrrrr-ZAP! The screw bites, the wood gives way, and suddenly, you’re a DIY hero. What gives? It’s not just about the quality of the bit, though that helps, believe me. It’s fundamentally about what that bit, and that screw, and frankly, almost everything around us, is made of.
And that, my friends, is our not-so-subtle segue into the wonderful, weird world of metals and nonmetals. They’re the rock stars and the quiet achievers of the periodic table, each with their own unique personality and job to do. Let’s dive in, shall we?
The Shiny, Bendy, Zappy Crew: What Are Metals?
Think about that trusty drill bit, or the pan you cook your eggs in, or even the jewelry you wear. What do they have in common? They’re likely metals. These guys are the popular kids of the material world, and for good reason.
First off, metals are usually shiny. We call this property lustrous, which is just a fancy way of saying they reflect light like nobody's business. Think of a polished silver spoon or a gold ring – bling factor 10!
But it's not just about looking good. Metals are also incredibly malleable. This means you can hammer them into thin sheets without them breaking. Ever seen gold leaf? That's gold, hammered so thin it's almost transparent. Pretty neat, huh?
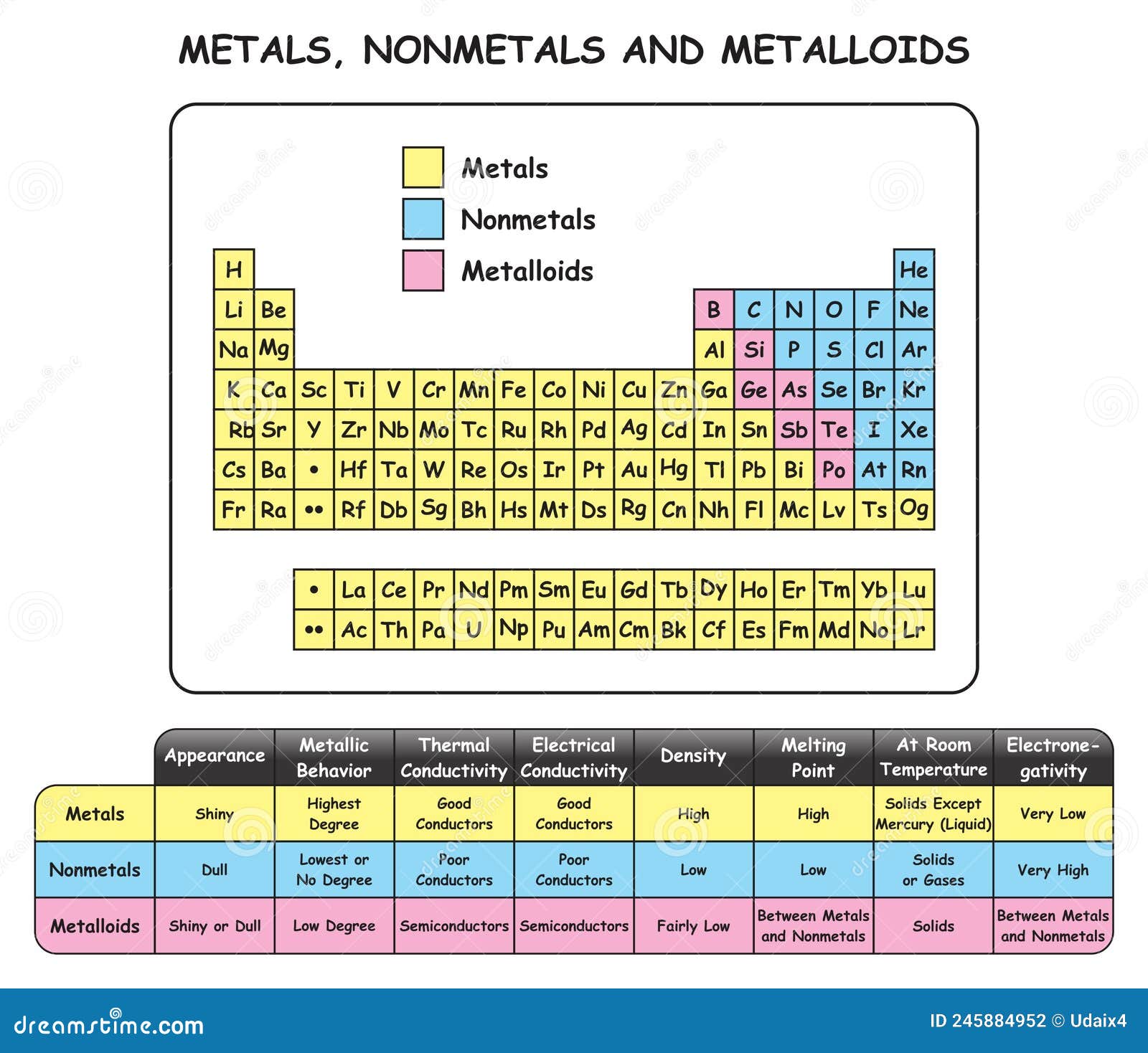
They're also ductile, which means you can pull them into wires. All the copper wiring in your house, your phone charger, even the tiny filaments in old lightbulbs (remember those?) – that's ductility in action. It’s why your devices can even exist, honestly.
And here’s a big one: metals are fantastic conductors of both heat and electricity. That's why your frying pan gets hot so quickly (and why you need a nonmetal handle, more on that later!). And it's how all your electronics work – electrons zip through metals like they’re on a superhighway.
Most metals are solid at room temperature (except for mercury, which is a liquid and frankly, a bit of an enigma), and they tend to have really high melting points. So, yeah, your car won't spontaneously melt on a hot day, thankfully.

The secret to all this awesomeness? Metals have a peculiar atomic structure where their outer electrons aren't tied down to individual atoms. Instead, they form a "sea of electrons" that moves freely. It’s like a giant party where everyone can roam around, which explains why they're so good at conducting electricity and heat, and why they're so flexible!
The Dull, Crumbly, Chill Bunch: What Are Nonmetals?
Now, let's swing to the other side of the periodic table, where the introverts of the material world reside. Nonmetals are essentially the opposite of metals in most ways, but they are no less important. In fact, you literally couldn't live without some of them (hello, oxygen!).
Unlike their flashy counterparts, nonmetals are typically dull, meaning they don't reflect light much. Think of carbon (like in coal or pencil lead) or sulfur. Not exactly the stuff of bling, right?
If they're solid, nonmetals are usually brittle. Try to hammer a lump of sulfur, and it’ll just shatter into a thousand pieces. Not quite as cooperative as gold. (Don't try this at home, kids, unless you like a mess.)
And remember how metals were super conductors? Nonmetals are generally insulators. They don't let heat or electricity pass through easily. This is why the handles on your metal pots and pans are made of plastic or wood – nonmetals keep your hands from getting fried! Their electrons are homebodies, tightly held by their atoms, not keen on traveling.
Nonmetals are also a diverse bunch when it comes to their state at room temperature. You’ve got gases like oxygen and nitrogen, a liquid like bromine, and solids like carbon and sulfur. Talk about variety!

The Awkward Middle Children: Metalloids
Just when you thought you had it all figured out, nature throws a curveball. Right in between metals and nonmetals on the periodic table, there’s a small group called metalloids. These guys are the fence-sitters, exhibiting properties of both metals and nonmetals.
Silicon, for example, looks a bit shiny like a metal but isn't as good at conducting electricity. However, it's good enough to be the backbone of virtually all our computer chips and electronics. Without metalloids, your smartphone would be a fancy paperweight, if it even existed at all. So, they might be awkward, but they're absolutely essential!
Why Should We Even Care, Right?
So, why all this talk about shiny things and crumbly bits? Because understanding the difference between metals and nonmetals is key to understanding the world around you. From the steel beams holding up skyscrapers to the oxygen you breathe; from the microchips in your phone to the plastic casing protecting it – it’s all about these fundamental differences.
Next time you pick up a tool, cook a meal, or even just breathe, take a moment to appreciate the incredible, diverse properties of the elements that make up our universe. It’s pretty wild when you think about it, isn’t it?