
Alright, gather 'round, folks, because today we're diving into the fabulous world of metals! Forget the boring science stuff; we're talking about the rockstars of the material universe. These aren't just any old lumps; they're the unsung heroes holding our world together, and they've got some seriously cool tricks up their sleeves. Get ready to have your socks charmed right off!
Metals are everywhere you look, from your sparkling jewelry to the mighty beams of skyscrapers. They make our lives easier, brighter, and a whole lot more exciting. So, what makes these metallic marvels so special?
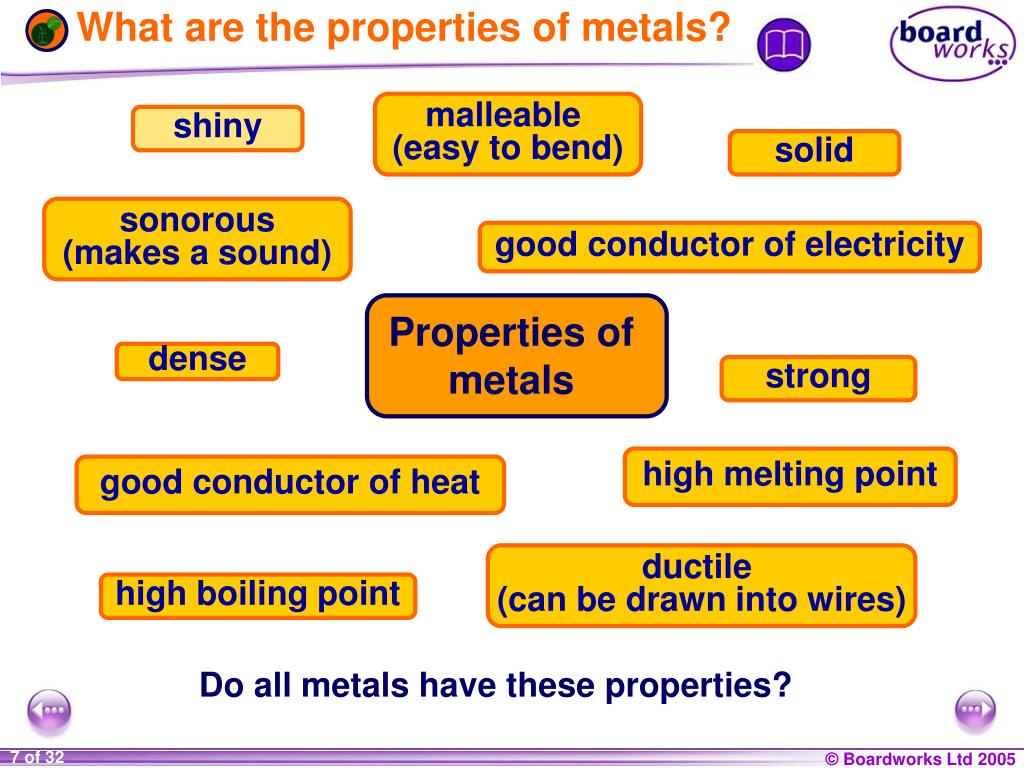
The Shiny Superstars: Lustre!
First up, let's talk about that undeniable sparkle. Metals, when they're fresh and clean, have this amazing ability to reflect light. We call this property lustre, and it's basically their natural superpower for shining brightly.
Think about a gleaming silver spoon or a polished gold ring. They just catch the light and practically scream, "Look at me!" This isn't just vanity; it's a fundamental characteristic that makes metals instantly recognizable.
Imagine a brand-new car, fresh off the lot. That dazzling paint job, often with metallic flecks, just shouts premium, doesn't it? That's the power of lustre at play, making things look fantastic and expensive.
No wonder we adore metallic accessories and shiny gadgets. They're just naturally appealing to our eyes! Metals truly are the ultimate masters of making a grand, glittering entrance wherever they go.
Hammer Time! Malleability and Ductility
Now, prepare to be amazed by metals' incredible flexibility. Unlike brittle ceramics or grumpy rocks, metals are surprisingly cooperative when you want to change their shape. They're like the yoga masters of the material world!
The Squish-and-Flatten Superpower: Malleability
Picture this: you hit a piece of metal with a really big hammer. Does it shatter into a million pieces? Absolutely not! Instead, it just gracefully flattens out. This amazing ability to be hammered or pressed into thin sheets without breaking is called malleability.
It's why we have things like super-thin aluminum foil to wrap your lunch. Or how artisans create intricate metal sculptures and car bodies that mold into sleek designs. Metals are so incredibly good at this; they practically beg to be reshaped!
Imagine trying to flatten a cookie or a stone into a sheet – total disaster, right? But metals? They're like, "Bring it on!" This property is a game-changer for manufacturing and artistry alike.

"Metals don't just take a hit; they transform into something new and beautiful!"
This fantastic ability means we can get incredibly creative with how we use them. From protective armor to decorative panels, metals are always ready for a dramatic transformation. They truly embrace the idea of change!
The Stretch-and-String Superpower: Ductility
Not only can metals flatten out, but they can also be stretched into unbelievably thin wires! This incredible trick is called ductility. It's like they're performing a magic act right before your eyes, turning solid chunks into slender threads.
Think about all the electrical wires powering your home and gadgets. Copper wires, thin as spaghetti, are responsible for carrying all that electrifying energy. Without ductility, our modern world would simply grind to a halt!
Gold, famously, is one of the most ductile metals. A single gram can be drawn into a wire miles long! It’s astonishing to think of a solid material stretching so far without snapping.
This property is also why you see metal fences, paperclips, and even delicate springs. Metals are just so incredibly versatile; they can be chunky one minute and gossamer-thin the next. They truly are the masters of transformation.
The Ultimate Connectors: Heat and Electrical Conductivity
Metals are not just pretty faces; they're also fantastic communicators. They're incredibly good at passing along both heat and electricity. This is where their superhero status really shines for everyday life.
Hot Stuff! Heat Conductivity
Ever wondered why your frying pan is made of metal? It's because metals are brilliant at conducting heat! They quickly absorb heat from your stove and spread it evenly across the pan, cooking your food perfectly. This property is called heat conductivity.
Imagine trying to cook with a wooden or plastic pan – disaster! Metals ensure your morning eggs get that perfect sizzle. They’re the ultimate heat delivery system, getting warmth exactly where it needs to be, quickly and efficiently.

This also means that if you grab a metal spoon left in a hot pot, it’ll get warm very fast. So, while fantastic for cooking, always use an oven mitt! Metals are just too efficient at their job.
They literally excel at moving energy around. This trait is essential for everything from radiators warming our homes to engines dissipating heat. Metals are the silent, efficient heroes of temperature transfer.
Zap-Tastic! Electrical Conductivity
Now for the really electrifying part: metals are phenomenal conductors of electricity. They basically act as superhighways for tiny electrical charges to zoom through. This is why all our wires are made of metal, often copper!
Without metals, your phone wouldn't charge, your lights wouldn't turn on, and your computer would be just a fancy paperweight. Electrical conductivity is the backbone of our entire electronic world.
"Metals are the unsung heroes powering our entire interconnected universe!"
They allow electrons to flow freely, making them indispensable for circuits, batteries, and everything that needs a good zap. Just think of the sheer volume of electricity coursing through metal wires every single second across the globe!
This incredible ability makes metals truly indispensable for modern technology. They ensure our gadgets stay charged and our homes stay lit, connecting us to the world at the speed of light.
The Mighty Weights: Density
Most metals are pretty hefty for their size. If you pick up a metal object, you’ll usually notice it feels quite substantial, sometimes surprisingly heavy. This is due to their high density.
Density simply means there's a lot of "stuff" packed into a small space. Metals cram in a lot of atoms, making them feel solid and weighty. This is why an anchor made of metal can hold a giant ship in place.
Think about how heavy a dumbbell is, even if it's not huge. That's density in action! It gives metals a certain gravitas and makes them excellent for building strong, stable structures.
While there are exceptions like lightweight aluminum, generally, when you think metal, you think solid and significant. They have a physical presence that just screams strength and reliability.
Tough Cookies: High Melting Points
Metals are not easily persuaded to turn into liquid. Most of them require incredibly high temperatures to melt down. This property is called a high melting point.
You can't just leave a metal spoon out in the sun and expect it to puddle! It takes serious heat, often in industrial furnaces or blacksmiths' forges, to get them molten. This makes them incredibly useful for things that need to withstand heat.
Imagine trying to cook in a pan that melted on the stovetop! Not ideal, right? Metals stay solid and reliable even when things get super hot.
This characteristic is vital for engines, industrial equipment, and anything exposed to extreme temperatures. Metals are truly tough cookies that stand firm against the heat, making them perfect for challenging environments.
The Bodybuilders: Strength
Metals are the ultimate bodybuilders of the material world. They are incredibly strong and can withstand a lot of force without breaking or bending permanently. This is their amazing tensile strength.
Think about giant bridges spanning rivers, towering skyscrapers, or the tools in your toolbox. All rely on the immense strength of metals to do their job safely and effectively. They are incredibly durable.

They don't just give up easily; they hold their ground against incredible stresses and strains. This is why we trust them to form the skeletons of our most important structures and machines.
"When you need something to hold up, metals are always the reliable choice!"
Their ability to resist deformation makes them indispensable. Metals are the workhorses that support our modern world, ensuring everything from tiny screws to massive girders stays firmly in place.
The Singers: Sonority
Finally, let's talk about how metals sound. When you strike a metal object, it often produces a clear, ringing sound, rather than a dull thud. This property is known as sonority.
Think of the beautiful chime of a bell, the crash of cymbals in an orchestra, or even the satisfying clink of cutlery. Metals resonate and produce pleasing sounds when they vibrate.
This musical quality is unique and adds another layer to their incredible versatility. They don't just look good and work hard; they can also make beautiful music!
So next time you hear a bell ring, give a little nod to the amazing sonority of metal. It's just another one of their fantastic qualities that adds a touch of charm and melody to our lives.
So there you have it! Metals are truly extraordinary materials, boasting a dazzling array of properties that make them essential to our lives. From their shimmering lustre to their incredible strength and conductivity, they are the unsung heroes shaping our world.
Next time you spot a piece of metal, give it a little appreciative wink. You're looking at a true superstar of science, making magic happen every single day! Aren't metals just utterly fantastic?