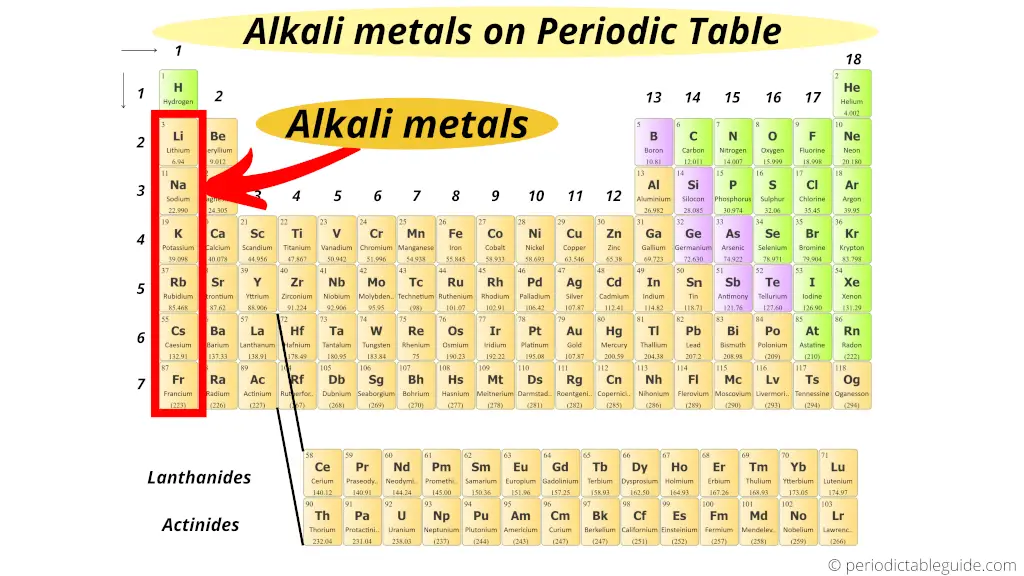
Imagine a family gathering where some relatives just have to be the center of attention. They’re loud, they’re dramatic, and frankly, a little high-maintenance. Well, welcome to the chemical world’s version of that family: the alkali metals. These shiny, soft, and unbelievably reactive elements make up Group 1 of the periodic table. And let me tell you, they’re quite the characters. Many people think they’re just too much trouble, too eager to react with anything. But I say, give them a chance! They’re just... enthusiastic. A bit like that one friend who turns every quiet coffee into an impromptu dance party. And honestly, isn't life a little more exciting with a few sparkling temper tantrums?
Lithium: The Energetic Toddler
First up in this lively bunch is Lithium. It's the smallest of the gang, often seen as the quiet one, but don't let that fool you. Lithium is still super reactive. It's like the quiet kid in class who, when finally given the microphone, totally shreds a guitar solo. We use Lithium in batteries, keeping our phones and laptops humming along. It’s also used in some medications, proving that even the most energetic elements can have a calming influence in the right dose. But try to drop a piece of Lithium into water, and you'll see a fizzing, bubbling, little chemical dance. It's pretty chill compared to its siblings, but it definitely wants to be noticed.
Sodium: The Flaming Torch Juggler
Then there’s Sodium. Ah, Sodium! This is probably the most famous, or infamous, alkali metal. You mostly know it from table salt, where it’s tamed down by pairing with chlorine. But by itself? Oh boy. Pure Sodium is a soft, silvery-white metal that you can actually cut with a butter knife. Sounds harmless, right? Wrong! Toss a chunk of pure Sodium into water, and you're in for a show. It zips around the surface, hissing and melting into a silver ball, often bursting into a brilliant yellow flame. It’s like the sibling who insists on juggling flaming torches at every birthday party. Some might call it reckless; I call it living life to the fullest.
"Why be subtle when you can be spectacular?" says Sodium, probably.
It just wants to share its inner fire with the world.
Potassium: The Pyrotechnic Pro
Moving on, we have Potassium. If Sodium is the lively teenager, then Potassium is its slightly wilder, older sibling who just discovered pyrotechnics. It's another soft, silvery metal, vital for our bodies (think bananas for good potassium!), but pure Potassium is even more eager to react than Sodium. Drop a piece of Potassium into water, and it reacts so violently that it usually ignites immediately, creating a beautiful lilac flame. There’s less zipping and more immediate fiery explosion. It doesn't just want attention; it demands it. I can almost hear it saying, "Hold my beer and watch this!" This dramatic flair is often seen as a downside, making it tricky to handle. But I think it’s just showing its passionate side. Who wants a boring element anyway?
Rubidium & Cesium: The A-List Divas
Now we're getting into the real divas: Rubidium and Cesium. These two are even further down Group 1, which means they are even bigger, and even more furiously reactive. They are so sensitive that they often react just with the air around them, bursting into flame. You usually have to keep them sealed in special glass ampoules under inert gas or oil, just to stop them from throwing a spontaneous fiery party. Imagine a celebrity so famous they need bodyguards just to go to the grocery store. That's Rubidium and Cesium.
Rubidium gives off a reddish-violet flame when it reacts, while Cesium – the true monarch of reactivity – produces a lovely blue-violet. Cesium is so incredibly reactive it will explode on contact with ice. Ice! Not even just water. It’s like it considers any molecule with oxygen or hydrogen an invitation to dance, vigorously. It’s used in atomic clocks because of its incredibly precise electron behavior, which is a surprisingly calm job for such a dramatic element. Some might call them "too hot to handle," but I prefer "intensely passionate." Their desire to bond with other elements is just unmatched.

Francium: The Vanishing Star
Finally, at the very bottom of the alkali metal family tree, we have Francium. This one is like the mythical, rarely-seen relative who only shows up in whispered legends. Francium is incredibly rare and intensely radioactive. It has a ridiculously short half-life, meaning it decays almost as soon as it forms. We know it should be the most reactive of them all, a true explosion-waiting-to-happen king, but it vanishes before we can truly get to know its full dramatic potential. It’s like the ultimate celebrity cameo: blink and you'll miss it. It probably wants to throw the biggest fiery party of all, but time just isn't on its side. Poor Francium.
So there you have them, the alkali metals. From the relatively mild-mannered Lithium to the elusive, explosive Francium. They are often dismissed as being too dangerous, too unstable, too much of a handful. They require careful storage and specialized handling, and yes, they'll probably try to ignite if you just look at them funny. But isn't there something admirable about such unbridled enthusiasm? About elements that are so eager to connect and transform? I think it’s time we gave these shiny, soft, and spectacularly reactive elements a little more love. They might be the drama queens of the periodic table, but they certainly make chemistry a whole lot more exciting. And honestly, who needs bland when you can have brilliant? My unpopular opinion? We need more sparkle in our lives, even if it comes with a tiny explosion.

