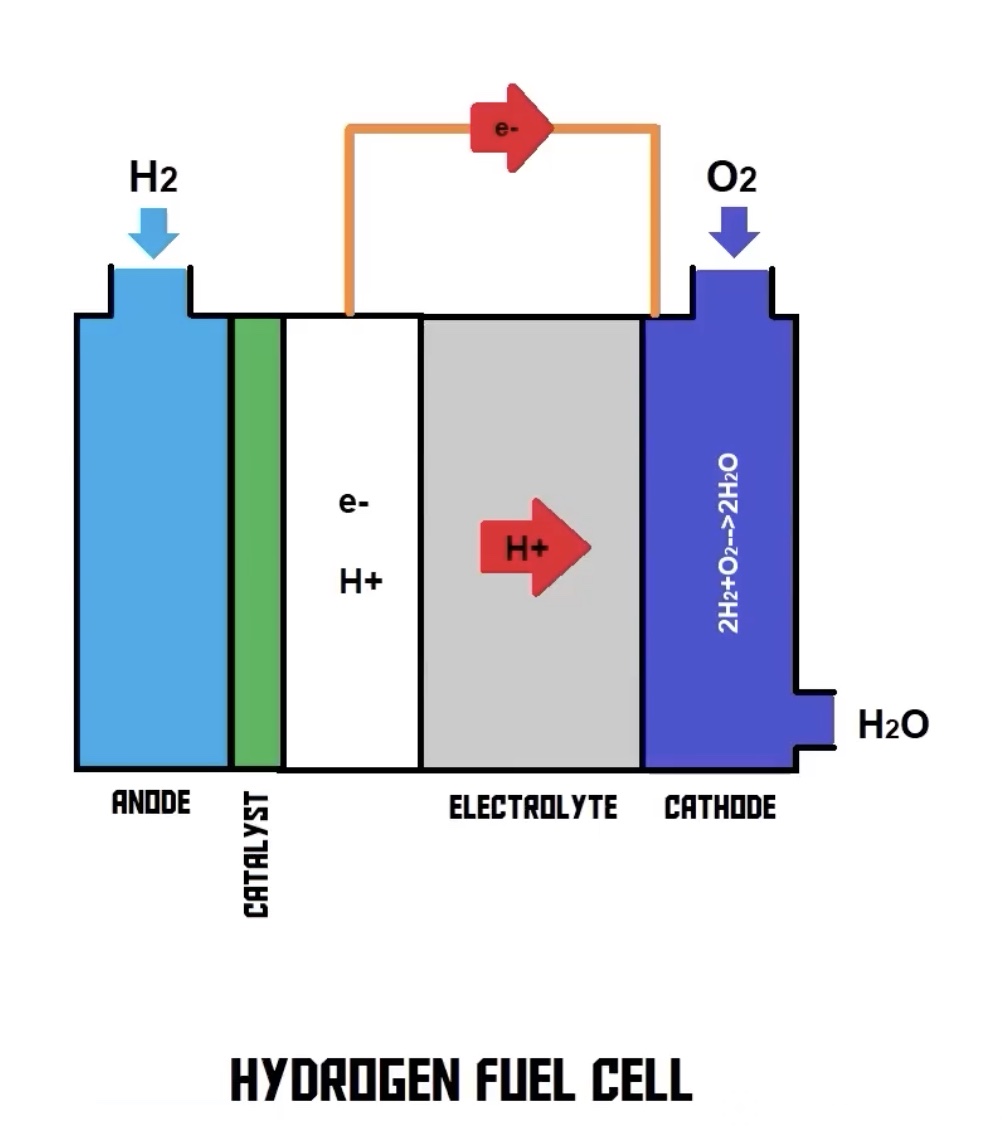
Have you ever stopped to think about the incredible magic happening inside the tech that powers our lives? It’s easy to just plug something in or fill up a tank, but sometimes, peeling back the layers reveals something truly mind-blowing. Today, we're going to peek behind the curtain of something super cool: the reaction inside a hydrogen fuel cell.
Forget complicated science textbooks for a moment. Let's just wonder together. Imagine a little box, a sort of energy wizard, that can take a gas and turn it directly into electricity, with almost no fuss and barely a whisper. That's essentially what a hydrogen fuel cell does!
What's the Big Idea, Anyway?
You’ve heard of batteries, right? They store energy and eventually run out, needing a recharge. A fuel cell is different. Think of it like a tiny, continuous power plant. It generates electricity as long as it's "fed" fuel – in this case, hydrogen. No recharging downtime needed, just a top-up of the good stuff.
And the "fuel" here is hydrogen (H₂). Yes, the most abundant element in the universe! It’s like having an almost limitless supply of potential energy just waiting to be tapped.
Meet Our Stars: Hydrogen and Oxygen!
For this show, we have two main characters:
- Hydrogen (H₂): Our eager, energy-packed hero. It's really just itching to give away its electrons and start a new adventure.
- Oxygen (O₂): The quiet, essential partner, waiting patiently to form something new. You know, the stuff we breathe!
When these two get together in a fuel cell, something really special happens. It’s not an explosion, not burning – it’s a calm, elegant dance that creates electricity.
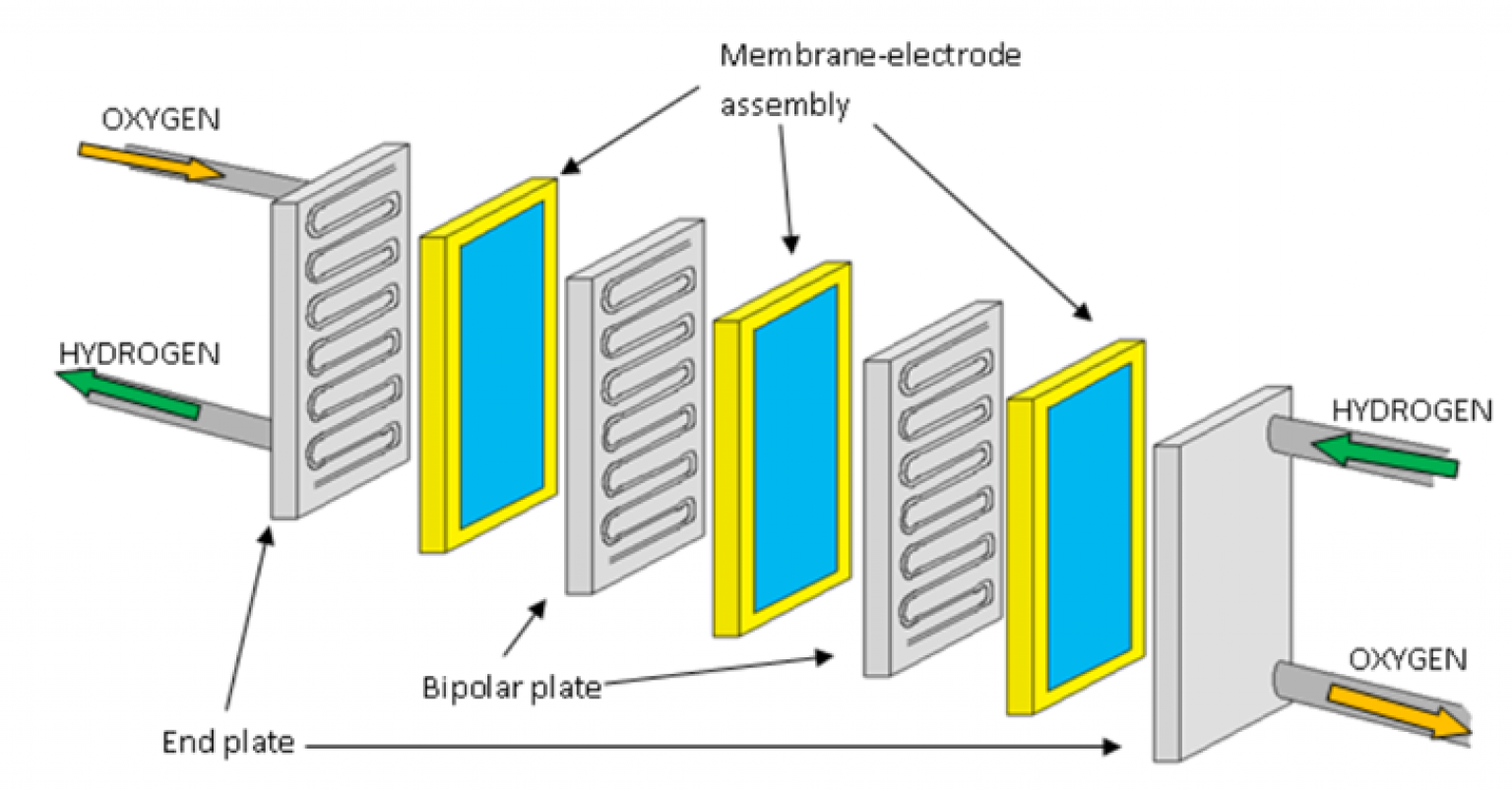
The Fuel Cell's "Magic" – A Step-by-Step Vibe
Step 1: Hydrogen's Grand Entrance (The Anode Side)
Imagine our hydrogen gas (H₂) entering one side of the fuel cell. It meets a special material called a catalyst, usually platinum (yes, the fancy metal!). This catalyst is like a super-friendly bouncer that helps hydrogen break apart.
When H₂ touches this catalyst, it does something extraordinary: it splits! Each hydrogen molecule (H₂) breaks into two hydrogen ions (H⁺) and two electrons (e⁻).
Think of it like hydrogen dropping off its baggage – its electrons – before going through a VIP door. These H⁺ ions are just hydrogen atoms minus their electrons, essentially protons.
Step 2: The Electron Escape! (Hello, Electricity!)
Now, those newly freed electrons (e⁻) can't go through the same VIP door as the hydrogen ions. Instead, they're forced to take a detour. This detour is what we call an external circuit.
As these electrons zoom around this circuit, perhaps to power a motor, light a bulb, or charge your phone, they are literally creating an electric current! This is the magic! This is the electricity we want!
So, in essence, hydrogen sheds its electrons, and those electrons go on a journey, doing work for us, before they can rejoin their hydrogen buddies. How cool is that?
Step 3: The VIP Pass (The Membrane)
While the electrons are out doing their thing, those hydrogen ions (H⁺) – our protons – are allowed to pass through a very specific gateway in the middle of the fuel cell. This gateway is called a proton exchange membrane (PEM). It's like a super-exclusive filter that only lets protons through, blocking everything else.
It ensures that the electrons have to go the long way around the external circuit, guaranteeing we get our power. Smart, right?
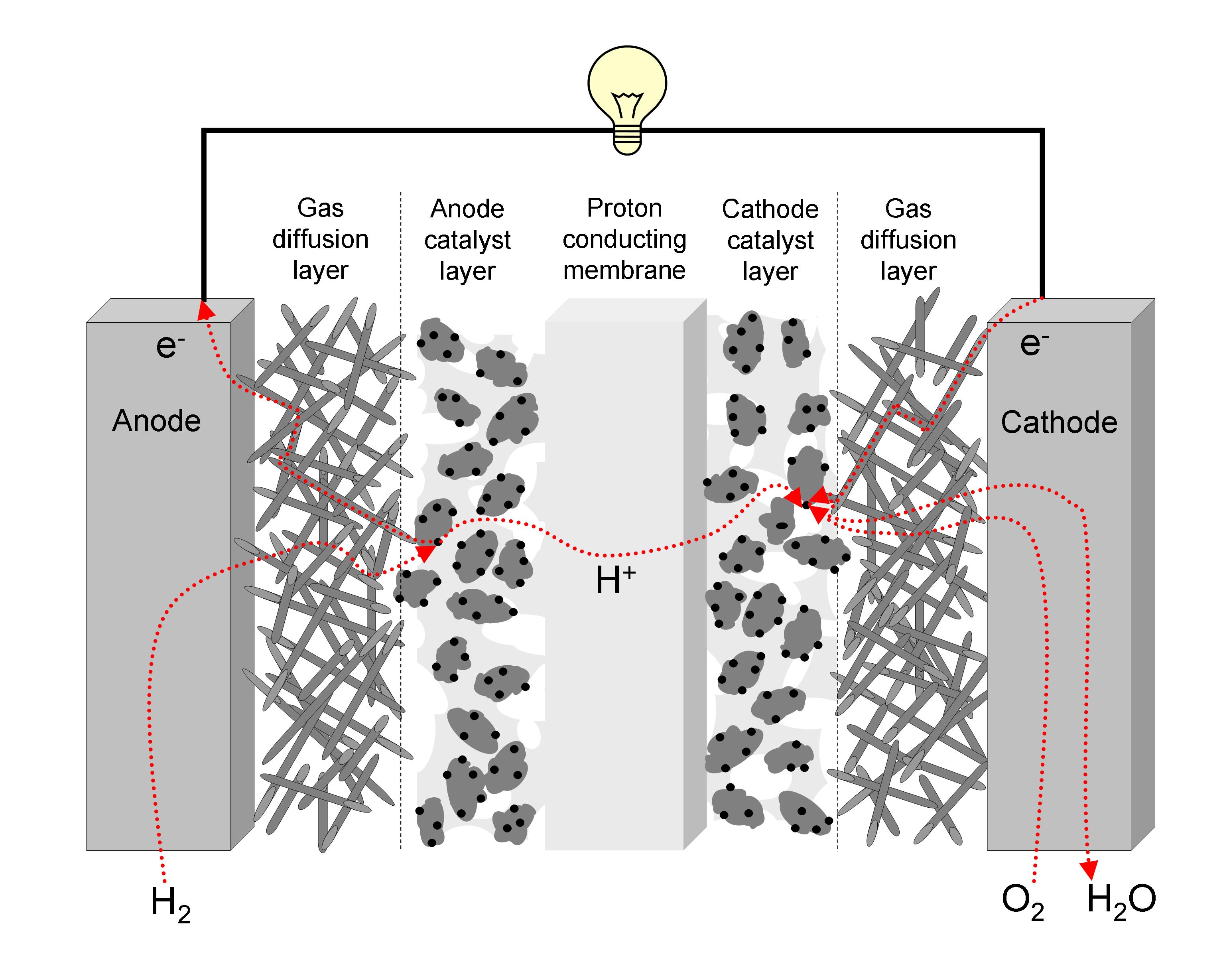
Step 4: The Grand Reunion (The Cathode Side)
On the other side of that membrane, our patient oxygen (O₂) is waiting. And guess who shows up? Those hydrogen ions (H⁺) that just passed through the membrane, and the electrons (e⁻) that just powered something awesome!
At this point, facilitated by another catalyst, the oxygen, hydrogen ions, and electrons all come together in a beautiful, simple chemical reaction.
What do they form? You guessed it: Water (H₂O)!
Why Is This So Awesome?
Let's recap:

- We started with hydrogen and oxygen.
- We got electricity out of it.
- The only byproduct is pure water!
Think about that for a second. No harmful emissions, no nasty fumes, just clean water dripping out. It’s like your car exhaling pure water vapor instead of exhaust! That's why fuel cells are considered such a clean energy solution.
And unlike combustion, where you burn fuel, this is an electrochemical reaction. It’s much quieter, more efficient, and directly converts chemical energy into electrical energy. It's a sophisticated, elegant process happening right inside that little box.
It’s not just a science experiment; it’s happening in cars, buses, even powering homes and data centers. The simple, elegant dance of hydrogen and oxygen creating electricity and water is a testament to the ingenious ways we can harness energy without harming our planet.
So next time you hear about hydrogen fuel cells, you'll know it's not some far-off sci-fi concept. It's a quiet, powerful, and incredibly clean chemical ballet happening right before your eyes (or rather, inside a very cool piece of tech)! Pretty neat, huh?