
Ever found yourself staring at a synthetic fabric – maybe your gym shorts, a sturdy tent, or even a toothbrush bristle – and wondered what on earth it’s made of? Chances are, you’re looking at nylon! And guess what? Nylon isn't just one thing. It’s a whole family, with two superstar siblings often taking the spotlight: Nylon 66 and Nylon 6. Sounds like secret agents, right? Let's dive into their super cool, slightly quirky structures without getting too tangled.
First off, what even is nylon? It's a type of plastic, specifically a polyamide. That fancy word just means it’s made of many repeating units (poly- meaning many) linked by something called an amide bond. Think of it like a long, atomic-level chain where each link is an amide bond. Super strong, super versatile!
Meet Nylon 66: The OG Powerhouse
Let's kick things off with Nylon 66. This is often considered the original, the pioneer, the one that DuPont chemist Wallace Carothers and his team famously cooked up in the 1930s. The "66" isn't just a catchy name; it's a secret handshake telling you exactly how it's built!
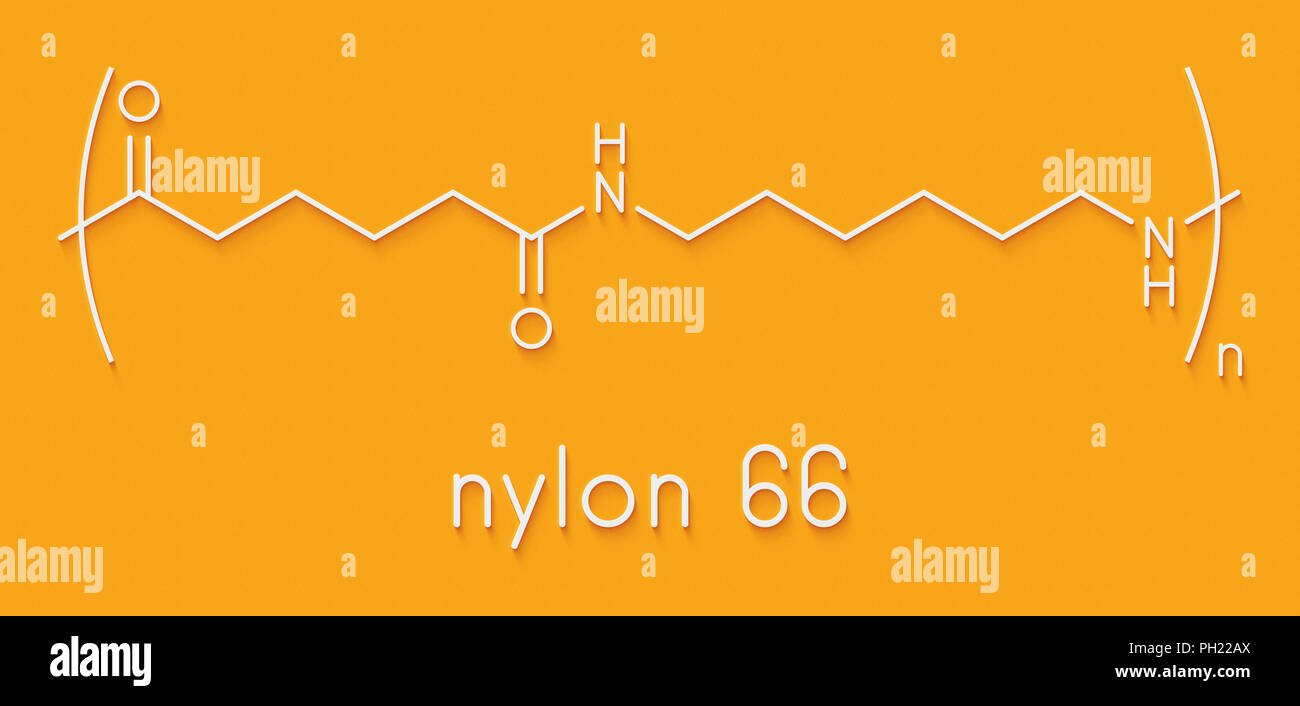
Imagine you're building with LEGOs, but instead of plastic bricks, you've got two specific types of molecular building blocks. For Nylon 66, you need two different "parent" molecules, and both of them happen to contain six carbon atoms. Yep, that's where the "66" comes from! One parent molecule is called hexamethylenediamine. Say that five times fast! It’s got six carbons and two amine groups (nitrogen-containing bits) on its ends, ready to make connections.
The other parent molecule is called adipic acid. And guess what? It also boasts six carbon atoms! This one has two acidic groups on its ends. So, you've got one molecule with two "hooks" (the amine ends) and another with two "loops" (the acid ends). When these two types of molecules meet under the right conditions – voilà! – they click together.

During this atomic click-and-connect process, something rather elegant happens: a tiny molecule of water is released for every bond formed. It's like the molecules are shedding water to hold hands! This process is called condensation polymerization. The result? A super long chain where hexamethylenediamine and adipic acid units alternate perfectly, creating a robust, crystalline structure. This specific architecture gives Nylon 66 its famous strength, stiffness, and high melting point. It’s why it’s great for tire cords, carpets, and those really tough fishing lines!
Now for Nylon 6: The Ring-Opening Rebel
Enter Nylon 6, the equally impressive but structurally distinct cousin. Its name, "6," is a clue that it’s made from just one type of monomer, and that monomer contains six carbon atoms. But here's where it gets quirky and fun!
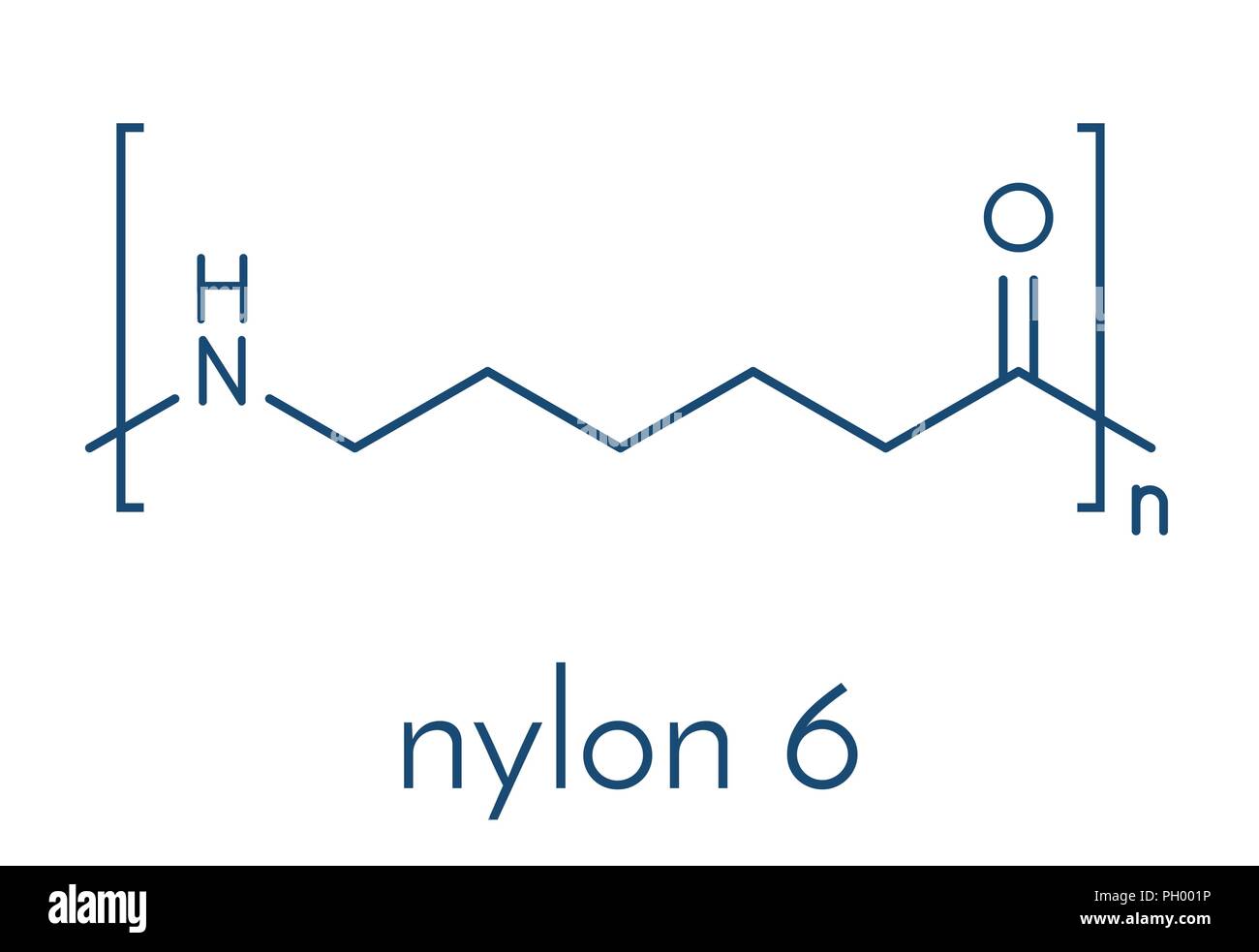
Instead of two separate LEGO bricks, Nylon 6 starts with a single, ring-shaped molecule called caprolactam. Imagine a snake that’s decided to bite its own tail, forming a perfect little six-carbon ring. Cute, right? Well, this ring is packed with potential. It has an amine group on one side and an acid group on the other, but they're inside the ring, silently waiting.
To make Nylon 6, you don't just "click" two molecules together. Oh no, that would be too simple! Instead, you take these caprolactam rings and, with a little heat and often a tiny bit of water or an initiator, you encourage them to break open. It’s like the snake deciding it's had enough of tail-biting and stretching itself out into a long, linear strand.
Once one caprolactam ring opens, its newly exposed "hook" and "loop" ends are itching to connect. They immediately grab onto the opened ends of other caprolactam molecules, linking up in a long, continuous chain. This is called ring-opening polymerization. It’s a totally different dance than Nylon 66's two-step tango, but it achieves a very similar result: a long, strong polyamide chain.
The beauty of Nylon 6's ring-opening trick is that it often makes it a bit easier to process. While both nylons are strong, Nylon 6 can sometimes feel a touch softer or more flexible, making it ideal for things like hosiery, apparel, and certain types of film and cast parts. Think of it as the slightly more pliable, but equally dependable, sibling.

Why Does This Matter?
So, why are these structural nuances so fascinating? Because these subtle differences in how their atomic LEGOs are arranged give Nylon 6 and Nylon 66 slightly different personalities and properties. While both are incredibly useful polyamides with excellent strength and resistance, one might be preferred for a super-tough industrial component, and the other for a comfy, durable textile.
They both share that fantastic amide bond backbone, which makes them resilient. But knowing that Nylon 66 forms from two distinct six-carbon monomers, and Nylon 6 from a single, ingenious six-carbon ring, adds a layer of appreciation for the cleverness of chemistry. It's like realizing that your favorite action heroes might have different origin stories, but both save the day with equal gusto!
Next time you pull on a piece of clothing or grab a synthetic kitchen tool, take a moment. You're not just holding "plastic"; you're holding a testament to molecular engineering, a marvel of tiny atomic structures doing big, impressive jobs. And that, my friend, is pretty cool.