Have you ever stopped to think about the everyday materials that shape our world? From the towering skyscrapers that grace our cityscapes to the humble spoon you use for your morning cereal, steel is undeniably everywhere. It’s strong, versatile, and incredibly useful. But here’s a fun little brain-teaser: if you were to grab a Periodic Table of Elements, would you find an entry for “Steel”? It’s a question that often sparks a moment of thoughtful pause, and exploring the answer gives us a delightful peek into the fascinating world of chemistry and materials science.
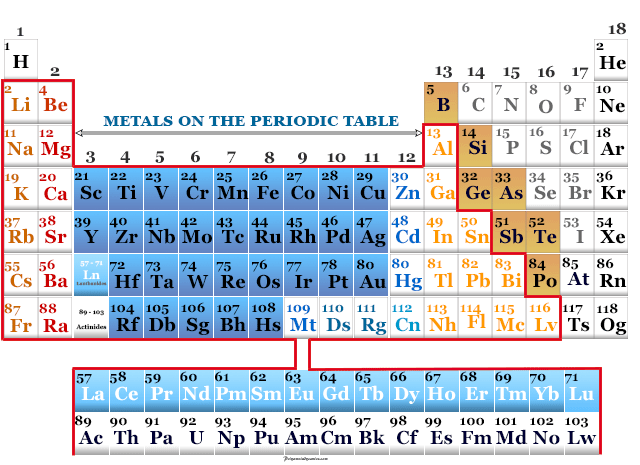
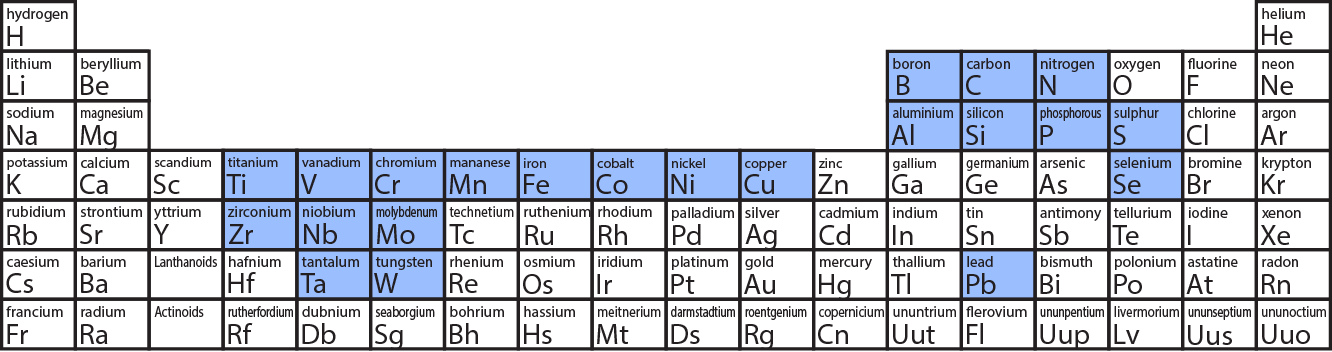
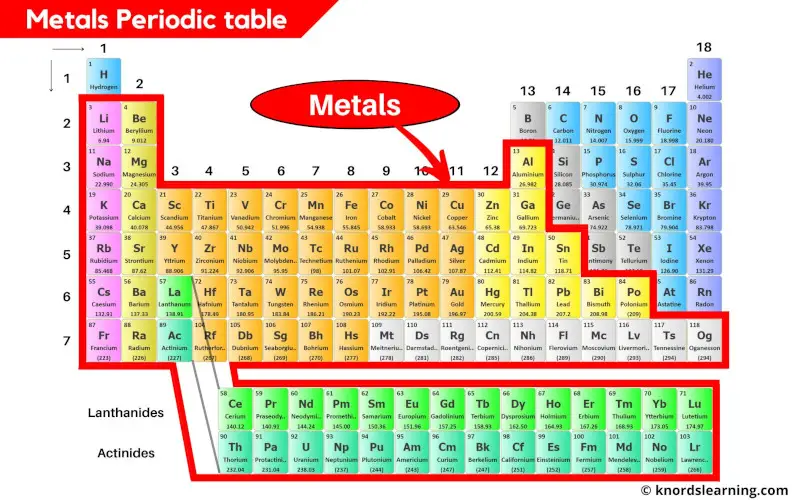
The Periodic Table is like the ultimate cheat sheet for the universe, listing all the fundamental building blocks – the elements – from hydrogen to oganesson. Each element is a pure substance, made up of only one type of atom, and they can't be broken down into simpler chemical substances. So, when we ask if steel is on the Periodic Table, we’re essentially asking if it’s a pure element. The answer, surprisingly to some, is a resounding no!
Steel isn't an element; it’s what we call an alloy. An alloy is a marvelous concoction, a mixture of two or more metals, or a metal combined with a non-metal, designed to have properties that are often superior to its individual components. In the case of steel, its primary ingredient is iron (Fe), which is an element found on the Periodic Table. To transform plain iron into steel, a small but crucial amount of carbon (C) – another element – is added, usually less than 2% by weight. Sometimes, other elements like chromium, nickel, or manganese are mixed in to create different types of steel, each with unique characteristics and specific benefits.
Understanding this distinction isn't just a quirky scientific fact; it's fundamental to appreciating the materials all around us. It teaches us that materials don't just exist as-is; they are often engineered and crafted by combining basic elements in precise ways. This knowledge is invaluable in fields like engineering, where choosing the right material for a bridge, a surgical instrument, or a high-performance engine is critical. In daily life, it explains why your stainless steel cutlery doesn't rust (thanks to chromium and nickel) while an old iron nail might (because it's mostly just iron). It highlights the ingenuity behind making materials stronger, lighter, more corrosion-resistant, or more flexible, simply by adjusting their atomic recipe.
So, how can you explore this concept further? Next time you see a steel object – perhaps a bicycle frame, a kitchen sink, or even the structure of a building – take a moment to ponder its origins. Remember that it began as iron, a silvery-grey element, and was then transformed into something incredibly useful by adding a tiny bit of carbon and perhaps other elements. You could look up different types of steel online, like "high-carbon steel" or "tool steel," and see how their compositions relate to their uses. You might even find an interactive periodic table and identify all the elements that make up your favorite stainless steel pan. It’s a simple way to connect the abstract world of chemistry with the tangible objects that fill our lives, turning an everyday observation into a moment of genuine scientific curiosity.