
Ever found yourself scrolling through a science documentary or maybe just pondering the periodic table while waiting for your coffee to brew, and a question pops into your head: Is carbon a metal or a metalloid? It’s one of those elements that feels like it’s everywhere, doing everything, from sparkling on your finger to being the very backbone of life. So, let’s peel back the layers on this elemental superstar, shall we?
The Great Carbon Conundrum: Metal, Metalloid, or Something Else Entirely?
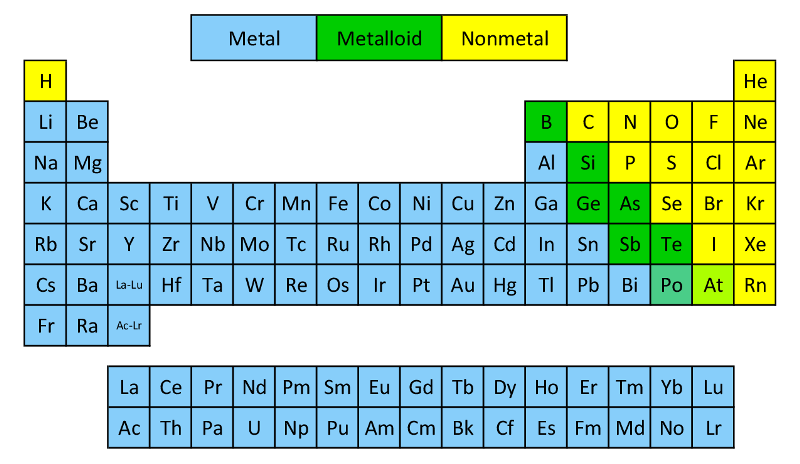
First off, let’s get into what typically defines a metal. Think about it: metals are usually shiny, good at conducting electricity and heat, and pretty malleable (you can hammer them into shapes) and ductile (you can pull them into wires). Gold, copper, iron – they all fit this description perfectly. They’re the rockstars of conductivity and durability.
Then we have the metalloids. These are the fascinating in-betweeners, like the chameleons of the periodic table. They exhibit properties that are a mix of both metals and nonmetals. Silicon, for example, looks a bit metallic but isn't as good a conductor as a metal. It's an excellent semiconductor, making it vital for all our tech gadgets.
So, where does carbon fit into this neat little classification? Many people might instinctively lean towards metalloid because carbon is so versatile and, in some forms, can conduct electricity. But here’s the scoop: carbon is actually a nonmetal.
Surprised? You’re not alone! While it sits right next to the metalloids on the periodic table (group 14, period 2), carbon doesn't typically share the properties of metals or even most metalloids. Most nonmetals are poor conductors of heat and electricity, lack luster, and are often brittle in their solid form.

However, carbon is anything but typical. It’s a true maverick!
Carbon's Amazing Allotropes: The Ultimate Shapeshifter
The reason carbon often throws people off is because of its incredible ability to exist in different structural forms, called allotropes, each with wildly different properties. This is where it gets really interesting:

- Diamond: The epitome of luxury and hardness. Diamonds are insulators, meaning they don't conduct electricity. They're also incredibly strong and lustrous. A clear nonmetal property here!
- Graphite: Soft, slippery, and dark, graphite is what makes your pencil write. And guess what? It’s a great conductor of electricity! This metallic-like property is due to its unique layered structure, where electrons are free to move. But despite this, it's still classified as a nonmetal because it lacks other key metallic traits like malleability.
- Graphene: The superstar of modern materials science! Graphene is a single layer of graphite, incredibly strong, flexible, and an even better electrical conductor than copper. It’s revolutionizing everything from electronics to biomedical tech.
- Amorphous Carbon: Think charcoal or soot. These are soft, black, and non-conductive, clearly nonmetallic.
So, while graphite and graphene might trick you with their conductivity, the overall picture of carbon, especially its common forms and chemical reactivity, firmly places it in the nonmetal camp. It loves to form bonds with other nonmetals, especially oxygen, hydrogen, and nitrogen, which is literally the basis of all organic life!
Why Carbon Matters: More Than Just Sparkles and Pencils
Understanding carbon's true nature isn't just a fun fact for your next trivia night; it helps us appreciate the world around us. Because carbon is a nonmetal, it readily forms a huge variety of compounds, making it the linchpin of organic chemistry and, by extension, all life on Earth.
Practical Tip: When you're choosing a non-stick pan, you might appreciate the carbon-based polymers that make cooking easier. Or when you pick up a super-light carbon fiber bike frame, you're experiencing the strength of this unique nonmetal.
From the carbon dating that tells us the age of ancient artifacts to the carbon dioxide we exhale and the graphite in our batteries, carbon is everywhere. It’s the ultimate connector, silently orchestrating the chemistry of our planet and our bodies.
A Little Reflection on the Elemental Wonders
So, the next time you marvel at a diamond’s sparkle, jot down a note with a pencil, or even just breathe, take a moment to appreciate carbon. It’s not a metal, nor is it a metalloid, but a truly unique and indispensable nonmetal that makes our world – and us – possible. It reminds us that sometimes, the most important things in life don't fit neatly into categories, and that's precisely what makes them so extraordinary.
