Alright, gather 'round, curious minds! Today, we're diving headfirst into a sparkling, shiny mystery that has tickled the brains of scientists and musicians alike for centuries. We're talking about brass, that shimmering champion of the metal world!
That glorious, golden-hued metal found in everything from gleaming trumpets to sturdy doorknobs. But here’s the million-dollar question that sparks our inner detective: Is brass a mixture or a compound?
Get ready for a fun ride, because we’re about to unravel this riddle with everyday examples that are way more exciting than a dusty old textbook. No need for a lab coat, just bring your biggest smile and your sense of wonder!
The Star of Our Show: Brass!
First, let’s take a moment to admire our protagonist, brass. It’s that beautifully warm, yellowish metal that just screams "classic" and "elegant," isn't it? It's celebrated for its impressive durability, its lovely shine, and its uncanny ability to make glorious music.
From the grandest symphony orchestra, where it powers the brass section, to the humblest antique shop, gracing ornate fixtures, brass is truly everywhere. Its versatility and aesthetic appeal are undeniable.
But what exactly is this golden wonder made of? Let's peel back its shiny layers and discover its fundamental components.
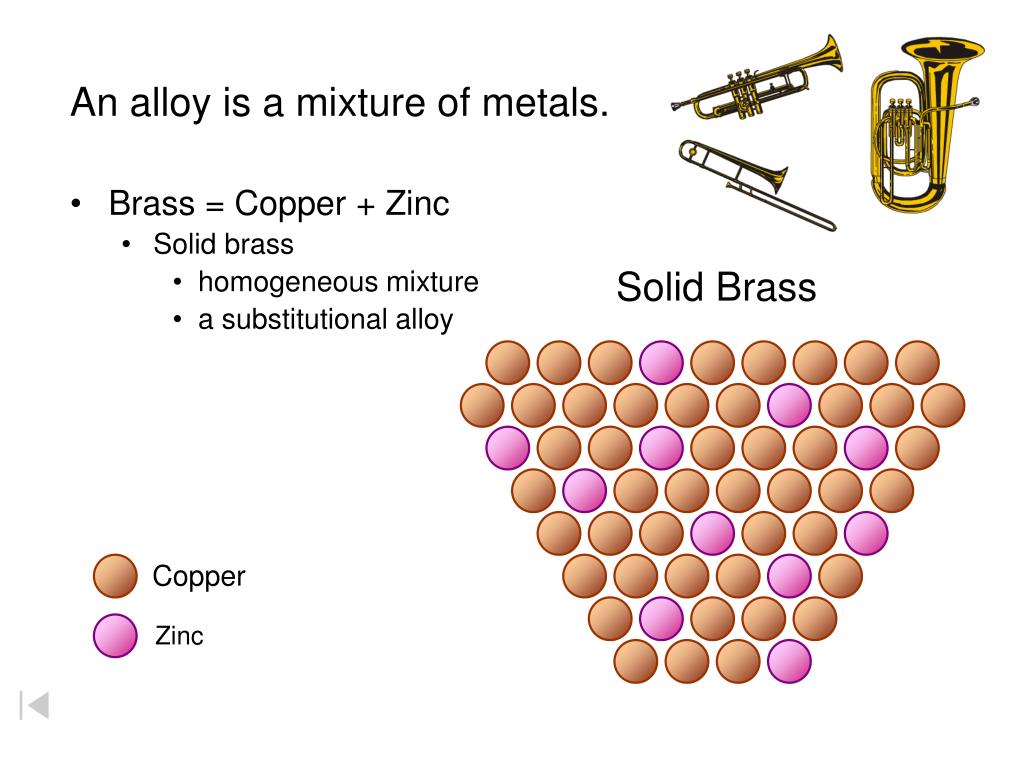
At its very heart, brass is primarily a harmonious combination of two other fantastic metals: copper and zinc. These two buddies are the secret ingredients behind brass's unique charm and widespread utility.
Mixture vs. Compound: The Epic Showdown!
What’s a Compound, Anyway?
Imagine two incredibly dedicated ingredients that decide to completely transform themselves, giving up their old identities entirely. They bond so intensely at a molecular level that they literally become a brand-new substance with profoundly different properties.
Think about water, for a perfect example. You start with hydrogen and oxygen, both gasses that you absolutely cannot drink. But when they join forces with a precise recipe, BOOM! You get liquid water, absolutely essential for life, and totally different from its gaseous parents.
Or take table salt, for instance! You start with sodium, a ridiculously reactive metal that would explode in water, and chlorine, a toxic green gas. But mix them just right, and suddenly you have delicious, harmless salt for your fries! That, my friends, is a powerful compound.
"Compounds are like chemical magic tricks – individual ingredients go in, and something utterly new and amazing comes out!"
With compounds, the ratio of ingredients is always meticulously fixed and incredibly precise. It's a very specific, unchanging recipe that cannot be easily altered or separated back into its original components without some serious scientific muscle and energy.
And What About a Mixture?
Now, picture a vibrant party where everyone shows up, still totally themselves, just happily hanging out together. They don't lose their individual identities; they simply co-exist side-by-side, mingling cheerfully. That’s a delightful mixture!
Think of a glorious fruit salad, bursting with color and flavor. You can still clearly see the bright strawberries, distinctively taste the sweet blueberries, and satisfyingly munch on the banana. They’re all there, enjoying the party together, but they haven’t magically turned into a giant, amorphous fruit goo.
Each piece of fruit keeps its own unique flavor, texture, and appearance. You can even painstakingly pick out your least favorite fruit if you wanted to! That's the undeniable beauty of a mixture – the individual components wonderfully retain their original properties.
Another fantastic example is a bag of trail mix, the ultimate snack party! You’ve got crunchy nuts, sweet chocolate chips, and chewy dried fruit, all living in delicious harmony. You can easily spot the peanuts, distinguish them from the M&Ms, and pick out the raisins without any trouble.

And here’s a crucial point for mixtures: the proportions can vary wildly, depending on who made it! One day your trail mix might be mostly nuts; the next, it's packed to the brim with chocolate. No strict recipe needed, just good vibes and a creative spirit!
Back to Our Golden Pal: Brass!
So, let’s apply these super-fun definitions to our shiny friend, brass. Remember, brass is made primarily of its two constituent metals: copper and zinc.
When copper and zinc get together to form brass, do they totally lose their identities? Does copper suddenly stop being copper? Does zinc magically transform into something utterly unrecognizable, shedding its metallic past?
Not quite! When you look at brass, you still clearly get a strong sense of its metallic nature, often reminiscent of copper's warmth. Zinc contributes its strength and its distinctive yellowish hue, but it doesn't create an entirely new, alien substance that's fundamentally different from both.
Brass essentially combines the most desirable properties of both copper (its gorgeous color, malleability, and electrical conductivity) and zinc (its added strength and a lower melting point). They work together beautifully, without one completely consuming the other into an entirely new, alien substance.
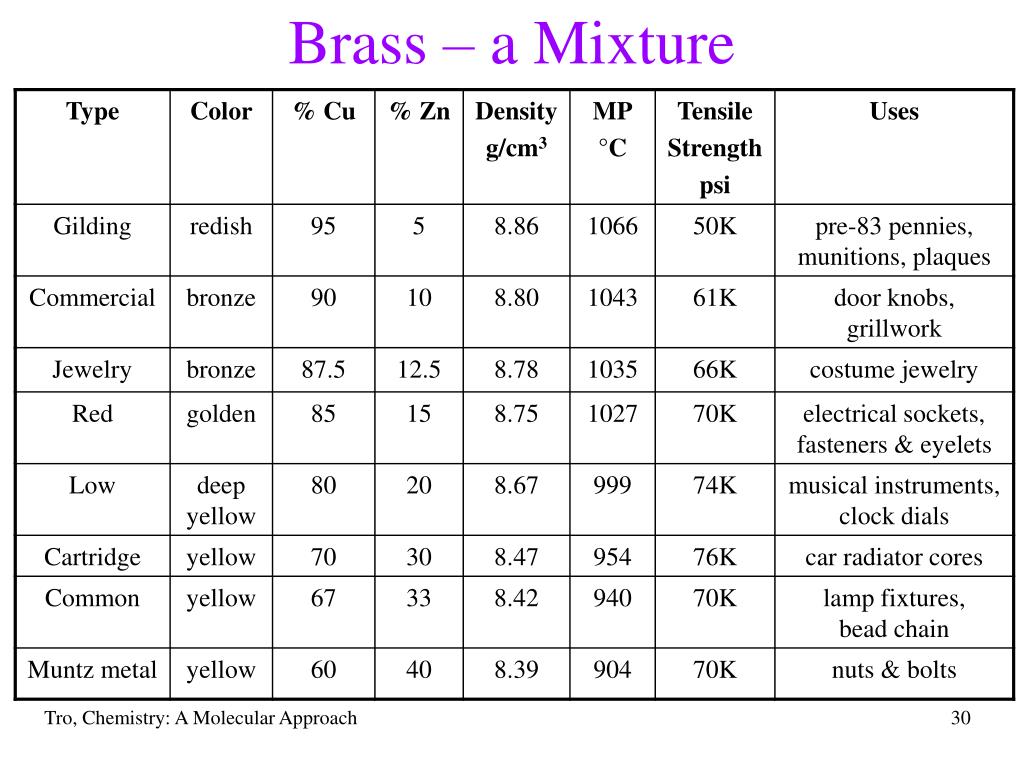
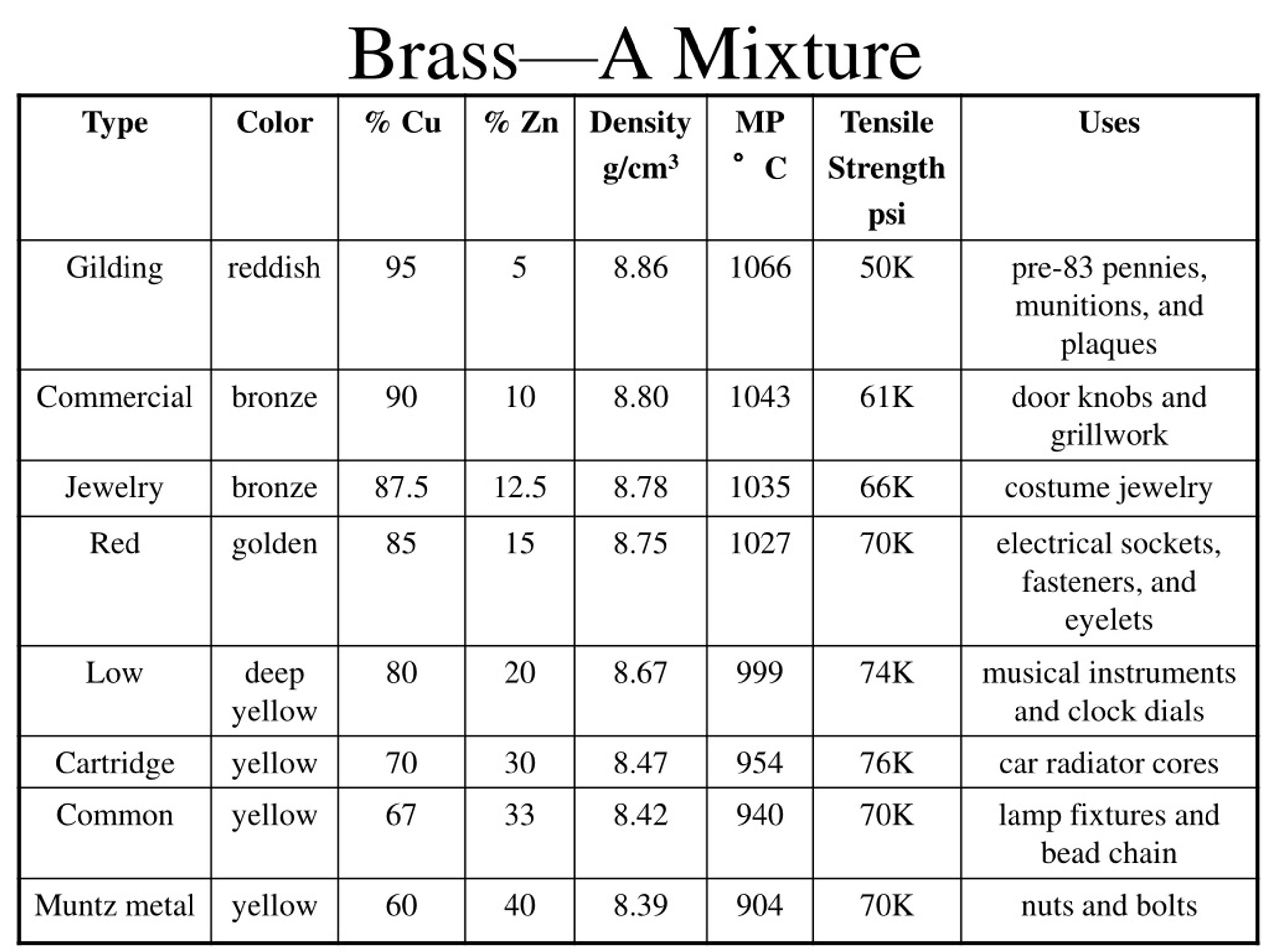
Now, let’s talk about those all-important ratios. Is the amount of copper and zinc in every type of brass always exactly the same, like the hydrogen and oxygen in water? Absolutely not!
There are many wonderfully different types of brass! You have red brass, which contains a higher percentage of copper and, predictably, boasts a lovely reddish tint. Then there's yellow brass, with more zinc, giving it a brighter, more pronounced yellow color.

This incredible variability in composition is a giant, flashing neon sign pointing us directly towards the answer. If you can playfully adjust the percentages of the ingredients and still confidently call it "brass," what marvelous truth does that unveil?
It tells us that copper and zinc are simply hanging out, like those fruits in a salad, or the goodies in a trail mix. They're blended together physically, not chemically fused into a brand-new entity with a strict, unyielding recipe.
You certainly can't easily change the ratio of hydrogen to oxygen in water without it stopping being water altogether. But you can absolutely play around with the copper and zinc ratios in brass, creating various, distinct varieties for different functional and aesthetic uses.
"Brass isn't a strict, unyielding recipe; it's more like a delicious culinary experiment where you can playfully tweak the ingredients to perfection!"
The Grand Reveal!
Drumroll, please! After all this delightful detective work, the answer is sparklingly crystal clear. Brass is, without a shadow of a doubt, a magnificent mixture!
Specifically, it's an alloy, which is just a super fancy word for a mixture of metals. It's essentially a solid solution where one metal (zinc) is uniformly dissolved or dispersed into another (copper), but they don't form new, irreversible chemical bonds that create a fundamentally different substance.
They wonderfully maintain their individual metallic characteristics, cleverly blending their strengths to create something even more useful and beautiful than either ingredient could be alone. It's truly a harmonious metallic blend, a testament to teamwork!
Why Does This Even Matter?
You might be thinking, "Who cares if it's a mixture or a compound, as long as my trumpet sounds good?" Well, understanding this is super cool because it helps us appreciate why brass behaves the way it does!
Because it's a mixture, we can precisely tailor its properties with incredible flexibility. Want a brass that’s super easy to machine for intricate parts? Simply add a little more zinc! Need one that's incredibly resistant to corrosion for marine environments? Adjust the copper content for superior resilience!
This incredible flexibility is precisely why brass is so utterly versatile and cherished in countless applications. From the intricate gears of a vintage clock, where precision is paramount, to sturdy plumbing fittings that need to withstand the test of time and resist wear, brass delivers!
It's not just a boring fact to memorize for a quiz; it's a key to unlocking the true magic of materials science. It allows brilliant engineers and visionary artists to custom-design brass for specific needs, making our world a more efficient, functional, and melodious place.
The Final Encore!
So there you have it, folks! The mystery is gloriously solved. Brass isn’t a tightly bound, new-identity-creating compound. It's a wonderfully blended, keep-their-cool-identities mixture.
A sophisticated alloy where copper and zinc mingle and enhance each other, without ever losing their fundamental metallic charm. It's like the ultimate metallic buddy system, bringing out the best in both!
Next time you see a gleaming brass object, give it a knowing nod and a wink. You'll now understand the simple, yet profound, chemistry behind its enduring shine and incredible versatility. Keep exploring, keep questioning, and always keep having fun with science!