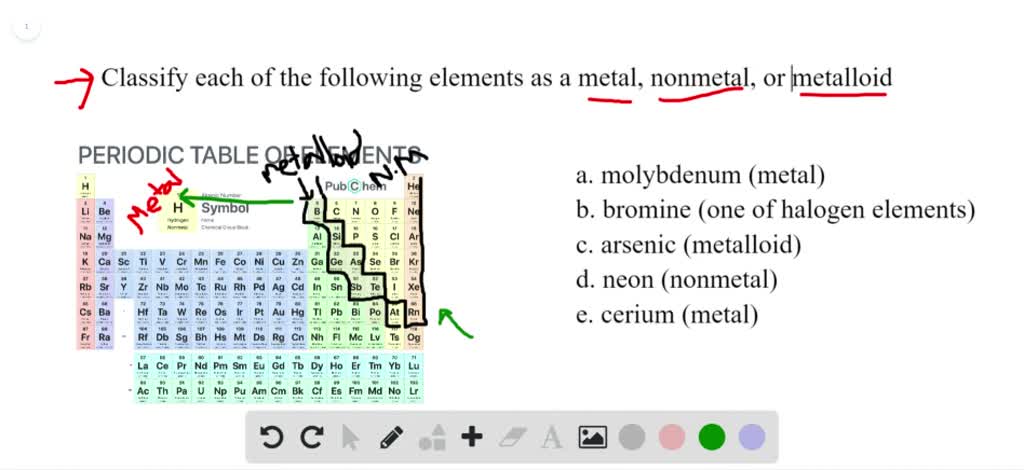
Ever paused to ponder the elements that make up our world? It’s a fascinating rabbit hole, especially when you stumble upon an element with a reputation like arsenic. We often hear its name whispered with a certain chill, but beyond the intrigue, arsenic holds a genuinely curious position in the chemical world. Understanding whether it's a metal or a nonmetal isn't just a quirky fact; it’s a delightful way to peel back the layers of the periodic table and appreciate the subtle complexities of matter itself.
So, let's get straight to it: is arsenic a metal or a nonmetal? The simple answer is neither, or perhaps, a bit of both! Arsenic belongs to a special group of elements called metalloids. Think of metalloids as the ultimate chemical fence-sitters, possessing properties that are intermediate between those of metals and nonmetals. This unique positioning is incredibly important because it gives arsenic, and other metalloids, distinctive characteristics that are highly valuable in various applications.
The real benefit of understanding elements like arsenic, especially their classification, lies in how it illuminates the entire field of materials science. It’s not just about memorizing categories; it’s about predicting how substances will behave, how they'll conduct electricity, or how they'll react with other elements. For metalloids, this means they can sometimes act like electrical conductors (a metallic trait) and sometimes like insulators (a nonmetallic trait), making them perfect for specialized roles. This deeper understanding helps scientists and engineers design everything from faster computers to more efficient solar panels.
In our daily lives and educational settings, the concept of metalloids often pops up. In chemistry class, when you first encounter the periodic table, you’ll notice a "staircase" line separating the metals on the left from the nonmetals on the right. The elements straddling this line – boron, silicon, germanium, arsenic, antimony, and tellurium – are our metalloids. Silicon, another famous metalloid, is the backbone of almost all modern electronics, from the smartphone in your pocket to the complex servers running the internet. Arsenic, in the form of gallium arsenide, is crucial for high-speed microelectronics, specialized lasers, and even some types of LEDs, thanks to its superior electronic properties compared to pure silicon in certain applications. Historically, arsenic compounds were used in pigments, pest control, and even some traditional medicines, though its high toxicity has led to a significant reduction in most of these uses today.
Want to explore this fascinating topic further? It’s surprisingly easy! Grab a copy of the periodic table (you can find countless versions online or in any science textbook). Look for the bold line, often a staircase pattern, that divides the table. Identify arsenic (element number 33, symbol As) and its metalloid neighbors. Notice how their position gives you a hint about their properties. You could also watch a few educational videos about "metalloids" or "semiconductors" on platforms like YouTube; there are many engaging explanations that bring these concepts to life. You'll quickly discover that the world of chemistry is full of delightful ambiguities and fascinating elements like arsenic, constantly challenging our simple classifications and inviting us to look a little closer.