Ever picked up a shiny soda can, admired a sleek bicycle frame, or marveled at an airplane, and thought, "Wow, aluminum is amazing!"? You're not wrong, but here’s a fun little secret that unravels a common misconception: What we often casually call "aluminum" is usually something a bit more sophisticated. It leads to a curious question: Is aluminum the same as an alloy? Let's dive into this metallurgical mystery that touches our daily lives, making everything from our kitchen utensils to high-speed transportation possible. It's a journey into materials science that's both fascinating and surprisingly practical!
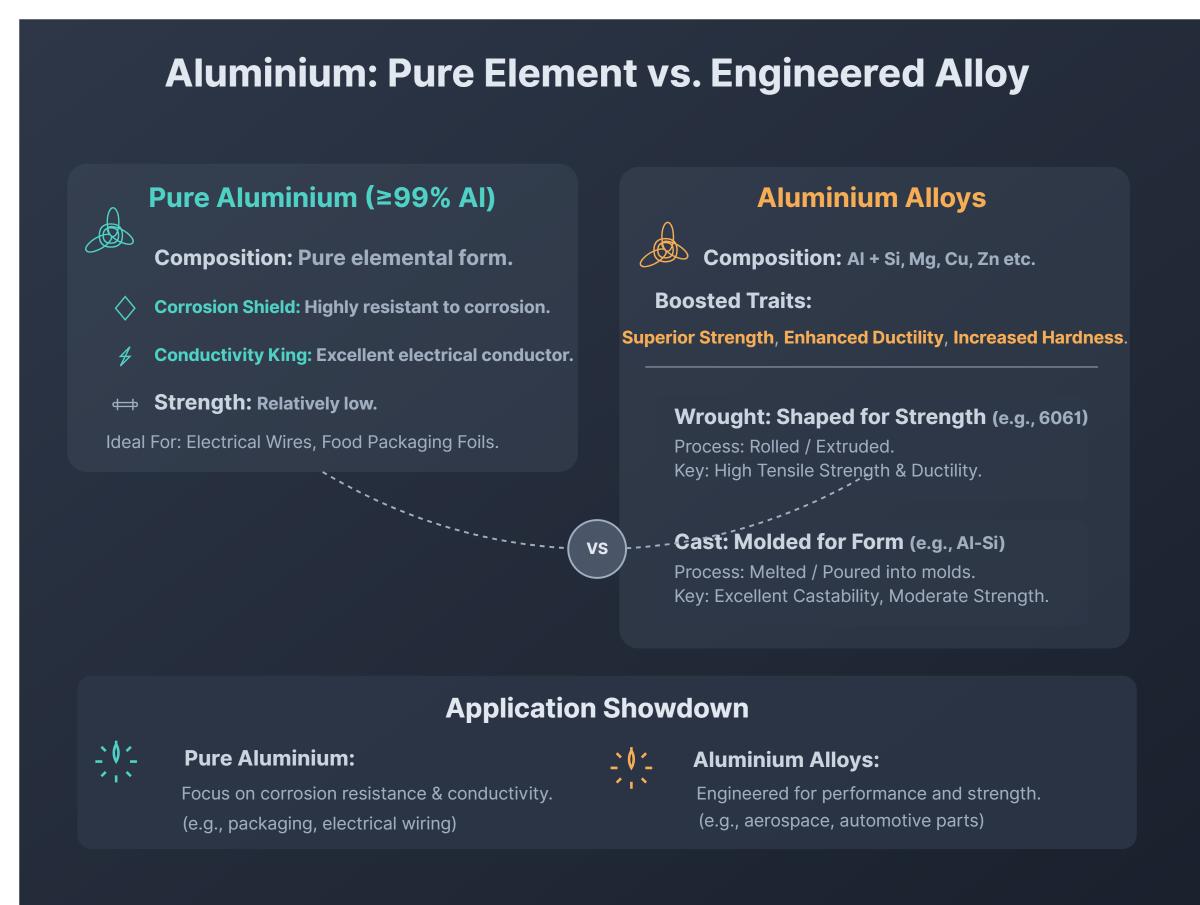
At its heart, aluminum is a pure chemical element, number 13 on the periodic table. In its pure form, it’s remarkably lightweight, has excellent electrical and thermal conductivity, and resists corrosion well because it forms a protective oxide layer. However, pure aluminum can be quite soft and malleable, which isn't always ideal for things that need to be strong or hold a specific shape under stress. This is where alloys come into play! An alloy is essentially a mixture of two or more metallic elements, or a metal and a non-metal, designed to create a new material with enhanced properties. The purpose is to combine the best characteristics of different elements, yielding a material that's stronger, harder, more durable, or even more resistant to specific types of wear than its individual components. So, the benefit is tailor-made performance for demanding applications.
Let's look at some everyday examples. While you might find nearly pure aluminum in things like very thin foil or some electrical wiring where its softness isn't a major issue, most of the "aluminum" you interact with is actually an aluminum alloy. Think about the frame of your mountain bike – it's incredibly lightweight yet strong enough to handle rough terrain. That's thanks to an aluminum alloy, often mixed with elements like magnesium, silicon, or zinc to boost its strength and stiffness. The soda can in your hand? It's an aluminum alloy, carefully engineered to be strong enough to withstand the pressure of carbonation, yet thin and lightweight. Airplanes rely heavily on advanced aluminum alloys (like Duralumin, containing copper) for their wings and fuselage, achieving the critical balance of strength and lightness necessary for flight. Even the pots and pans in your kitchen are typically aluminum alloys, offering better durability and heat distribution than pure aluminum.
So, how can you explore this fascinating difference yourself? Next time you're handling something made of "aluminum," take a moment to consider its properties. Is it bendy and soft like foil, or rigid and strong like a can or a bike part? You're likely experiencing the difference between nearly pure aluminum and an aluminum alloy. A simple tip: look at the labels on products or do a quick search online for terms like "aircraft-grade aluminum" or "aluminum alloy 6061" (a common alloy for bicycle frames). You’ll often find detailed information about the specific elements added and why. You could even compare a sturdy aluminum bottle to a piece of aluminum foil and ponder the forces that make one so much more resilient. It’s a subtle distinction, but understanding it gives you a deeper appreciation for the engineered materials that shape our modern world!