
Alright, settle in, grab your beverage of choice. We're about to dive into one of those questions that probably pops into your head every time you glance at a nutrition label, right between wondering if you really need that extra cookie and debating the meaning of "partially hydrogenated." The big one: how on earth do they know how many calories are in this croissant? Do they have tiny little food scientists with magnifying glasses, gently poking each crumb? Or is there some kind of mystical food oracle involved?
Prepare to have your mind slightly blown, because the answer, at least historically, is surprisingly dramatic and involves a lot of... fire.
The Original "Burn It All Down" Method
Back in the day, when scientists first got curious about how much energy our food held, they didn't mess around with complicated equations. Their logic was delightfully simple: if energy is heat, and food contains energy, then let's just burn the food and see how much heat it gives off!
I know, right? Imagine the lab accidents. "Oops, there goes another bagel!"
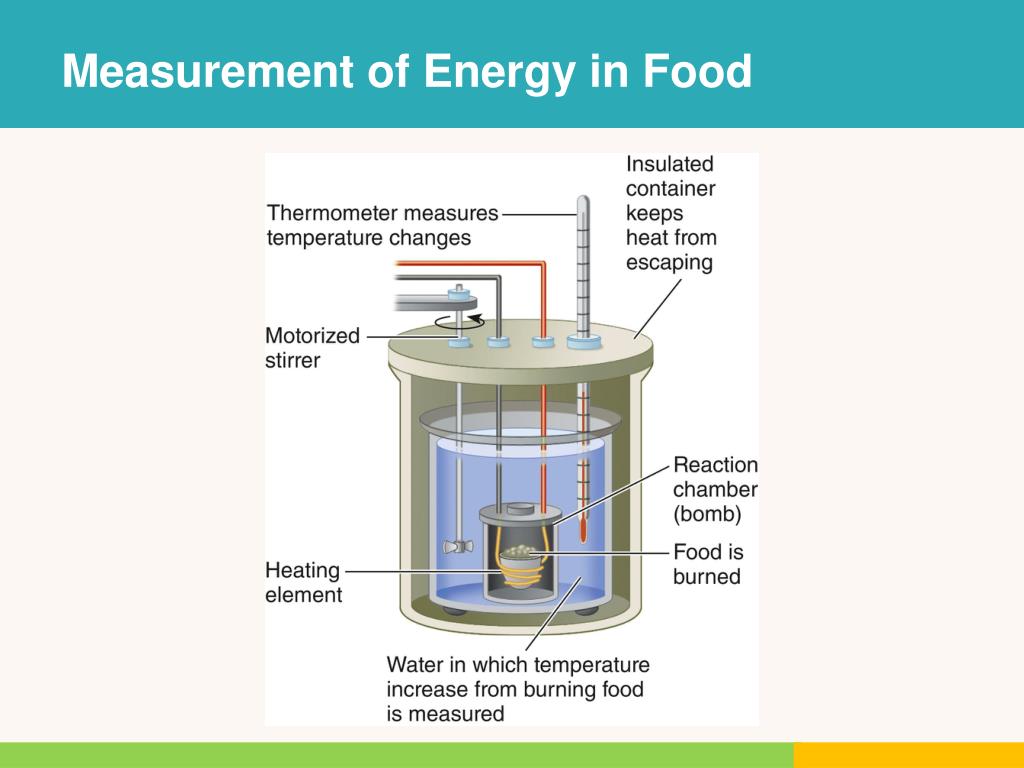
This wasn't just some guy with a lighter and a marshmallow, though. They used a contraption straight out of a mad scientist's workshop: the bomb calorimeter. Don't worry, it doesn't actually explode, unless you've somehow managed to stuff a small firecracker into your oatmeal.

Here's the gist: you take a precise amount of food, dry it out completely (because water just complicates things, bless its hydrating heart), and then pop it into a sealed, super-strong metal container. This container is then submerged in a known amount of water.
And then? Whoosh! Oxygen is pumped in, and the food is ignited. It burns completely, like a tiny, very scientific campfire. As the food incinerates, it releases heat, which warms up the surrounding water. By measuring the change in the water's temperature, scientists could calculate exactly how much energy was released. It’s like a very precise, slightly terrifying, gastronomic thermometer.
Every degree the water temperature rose told them how many calories (or kilojoules, if you're feeling fancy and metric) were packed into that humble bite.

Wait, Calories vs. "Calories"?
Quick clarification, because this gets confusing. When you see "Calories" on a food label (with a capital 'C'), what they actually mean is kilocalories (kcal). One kilocalorie is 1,000 "little c" calories. So, that 200-Calorie snack is actually 200,000 little-c calories. Why the confusing shorthand? Probably because "200,000 tiny calories!" sounds much more intimidating, and frankly, a bit ridiculous to say every time. So, we've just agreed to be cool with the capital C and know what we mean.
Our Bodies Aren't Incinerators (Thankfully)
While the bomb calorimeter gives a great theoretical maximum energy, here's the kicker: our bodies don't work like that. We don't just torch everything we eat into a puff of smoke. We digest, absorb, excrete. Some things, like fiber, pass right through us, barely contributing any usable energy, even though a calorimeter would happily set it ablaze.
So, while the burn-it-all method was a brilliant starting point, it wasn't quite accurate for predicting what our bodies actually get out of food.

Enter the Atwater System: The Smarter Way
This is where a clever chap named Wilbur Olin Atwater (yes, a real person, not a character from a Victorian novel) came in around the turn of the 20th century. He figured out that different components of food – carbohydrates, proteins, and fats – yield different amounts of usable energy once digested by a human.
He spent years conducting incredibly detailed experiments, measuring everything people ate and everything they... well, didn't absorb. It was painstaking work involving a lot of "input" and "output" analysis. His findings led to the famous "Atwater factors":
- Carbohydrates: Roughly 4 Calories per gram
- Proteins: Roughly 4 Calories per gram
- Fats: A whopping 9 Calories per gram
- Alcohol: (For those curious!) About 7 Calories per gram
Notice how fat packs more than double the punch? That's why a small amount of oil can quickly ramp up the calorie count. It's the energy dense superstar (or supervillain, depending on your diet plan).
Today, food manufacturers mostly use the Atwater system. Instead of burning every single batch of cereal, they calculate the calorie count based on the proportion of carbs, protein, and fat in the ingredients. It’s a lot less messy and probably reduces the risk of setting off the factory's smoke alarms daily.
Why the Numbers Aren't Always Perfect (And That's Okay)
Even with Atwater's genius, these numbers are still averages.
- Your body is unique: Your gut microbiome, how thoroughly you chew, and your individual digestive enzymes can all affect how many calories you actually extract from food.
- Cooking matters: Raw vs. cooked food can change digestibility. Ever tried to digest a raw potato? Don't.
- Rounding: Food labels are allowed a bit of wiggle room (often up to 20%) for rounding and variation. So, your "200 Calorie" snack might actually be 180 or 220.
So, the next time you look at that nutrition label, you'll know it's not just a random guess. It's the result of over a century of scientific curiosity, involving everything from tiny incinerators to meticulous digestive tracking. It's a blend of fiery experiments and careful calculations, all to help you understand the energy potential packed into your next bite. Pretty cool, right? Now, about that croissant...