
Hey there! Grab a coffee, settle in. Let's chat about something super cool from the periodic table, often overshadowed by its flashier relatives. You know, those elements?
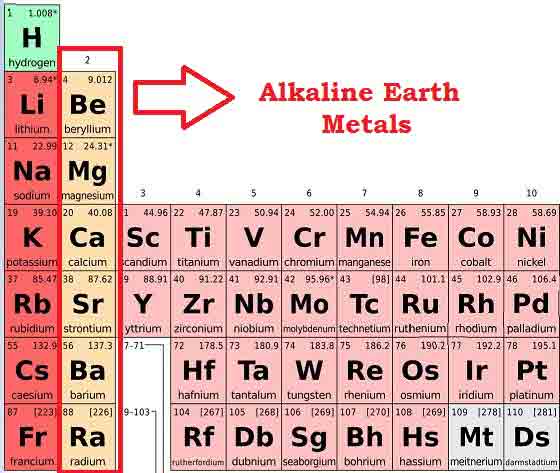
We’re talking about a particular bunch, sitting pretty in the second column. Group 2 metals. Ever heard of them? Maybe, maybe not. But here’s a little secret: they have a much cooler nickname. Like, their official street name, if you will.
The "Alkaline Earth" Vibe
Ready for it? Drumroll please… Group 2 metals are also called the Alkaline Earth Metals. Catchy, right? Sounds a bit like a band from the 70s, or a really chill yoga retreat. But why that name, you ask? Glad you’re curious!
The "alkaline" part? Their compounds often form solutions that are, yep, alkaline when they react. Think opposite of acidic. They're pretty good at neutralizing acids. Like little chemical superheroes, saving the day from too much sourness!
And "earth"? That's because they're commonly found right here in the Earth’s crust. Super abundant, making up a big chunk of our planet. They’re truly grounded members of the elemental community (pun totally intended).
Meet the VIP Family!
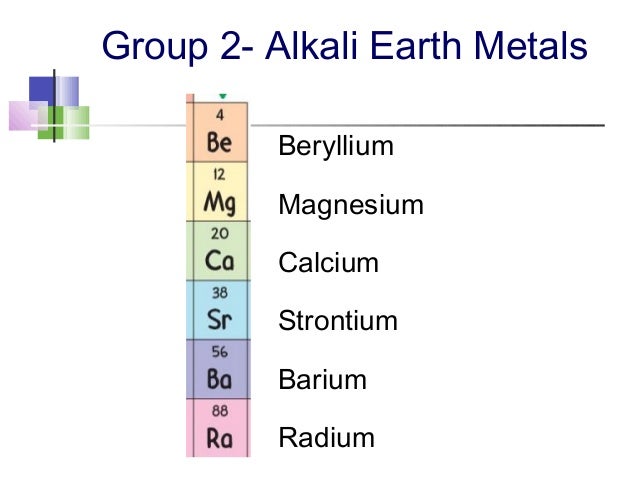
So, who are these magnificent creatures? Let’s meet the family! We’ve got Beryllium (Be), Magnesium (Mg), Calcium (Ca), Strontium (Sr), Barium (Ba), and the slightly infamous Radium (Ra). Each one has its own little personality, like a quirky family reunion.

Beryllium? Kinda shy, but super strong and light. The quiet, athletic one. Used in aerospace because, well, light and strong is a good combo for flying high!
Then there's Magnesium. Oh, Magnesium! A total star. Epsom salts? Yep, Magnesium sulfate. Green plants? Chlorophyll has a Magnesium atom right at its heart! Plus, fireworks use Magnesium for that super bright, white flash. Talk about versatile!
And Calcium. Ah, Calcium. Your bones love it. Your teeth love it. Milk commercials adore it. It’s the king of structural integrity in your body. Think chalk or concrete – big doses of Calcium compounds. Basically, Calcium is everywhere, helping things stand tall and strong.

Moving on, we have Strontium. This one’s a bit of a show-off, in a good way! Red fireworks? Thank Strontium! It gives them that gorgeous, vibrant crimson. Pretty cool party trick, right?
Next up, Barium. This one's more behind-the-scenes. Ever heard of a "barium meal" for an X-ray? That’s Barium sulfate, making your insides visible without being absorbed. A temporary internal highlighter!
And finally, Radium. Radium is the slightly rebellious, radioactive cousin. Famous for making watch dials glow (before people realized the… issues). We don’t really hang out with Radium much anymore, for obvious safety reasons. Best admired from a very, very safe distance. It adds a bit of danger to the family tree.
What Makes Them Tick?
Beyond their cool names and quirks, what's their big secret? Why are they all lumped together in Group 2? It all comes down to their electrons. Specifically, they all have two valence electrons. Think of these as their outermost, ready-to-party electrons.
These two electrons? They’re just itching to be given away. They want to be stable, and shedding those two is the easiest way. So, they easily form positive ions with a +2 charge. They're basically saying, "Take these! We don't need them! We just wanna be happy and stable!"
This desire makes them pretty reactive. Not as wildly reactive as Group 1 Alkali Metals (those guys only have one electron to lose, so they're even more desperate!). Group 2 metals are more like the calmer, but still very eager, siblings. They'll fizz and bubble, but usually without the dramatic explosions of Group 1. Phew!
They’re also generally shiny, silvery-white metals. Classic metal looks. But don't let their good looks fool you; they're hard at work behind the scenes.
So, What's the Big Deal?
Honestly, the big deal is how much they impact our everyday lives, often without us even thinking about it. From your bones to fireworks, from chlorophyll in plants to essential nutrients in your diet, these Alkaline Earth Metals are truly unsung heroes.
So, next time you see a bright white flash, or pour yourself a glass of milk, or just look at a green leaf, give a little nod to the Group 2 metals. Or, you know, their cooler alter ego: the Alkaline Earth Metals. They might not always get the spotlight, but they're definitely rocking our world, one two-electron donation at a time.
Pretty neat, right? Now, about that coffee… refill?