Alright, gather 'round, folks! Ever looked at a shiny brass trumpet, a fancy doorknob, or maybe even those snazzy bullet casings and thought, "Huh, what is this stuff?" It’s got that distinctive warm glow, right? Not quite gold, not quite copper, but something totally its own. And then, if you're anything like me, your brain probably wanders into the weird corners of science: Is brass a mixture or a compound?
It's a question that sounds super science-y, like something a professor in a tweed jacket would ask, but honestly, it’s as relatable as deciding what to eat for dinner. Seriously!
Let’s Talk Compounds: The Un-Unmixable
Imagine you're making a cake. You throw in flour, sugar, eggs, butter, maybe a dash of vanilla. Before it goes into the oven, you can still kinda see the eggy bits or specks of flour, right? But then, BAM! Heat. Chemical reactions. What comes out? A glorious, unified cake. You can't just pick out the raw flour or the separate eggs anymore. They’ve transformed.
That, my friends, is a compound. Think of water, H2O. You started with hydrogen gas and oxygen gas, two entirely different things. But when they get together, they don't just hang out; they form a super-tight, inseparable bond. Water looks nothing like hydrogen or oxygen on their own. It's a brand new entity, with new properties. It’s like a really committed, full-on merger where everyone loses their individual identity to become one super-entity. No take-backs!
Now, for Mixtures: The Chill Roommates
Now, let's swing to the other side: a mixture. This is where things are a bit more... casual. Think about a fruit salad. You’ve got apples, grapes, strawberries, maybe some melon. They're all in the same bowl, chillin' together, mingling. But can you still point to an apple slice and say, "Yup, that's an apple"? Absolutely! You can even pick out the grapes if you don't like them (weirdo).
Another classic: trail mix. Peanuts, M&Ms, raisins. You can literally pick out all the M&Ms first, because, priorities. The ingredients are combined physically, but they haven't chemically morphed into something new. They retain their individual properties. It's like a bunch of roommates sharing an apartment. They live together, share bills, maybe even a Netflix account, but they’re still distinctly themselves. They don't suddenly become one giant, multi-limbed roommate blob. Thank goodness.
So, Where Does Brass Fit In?
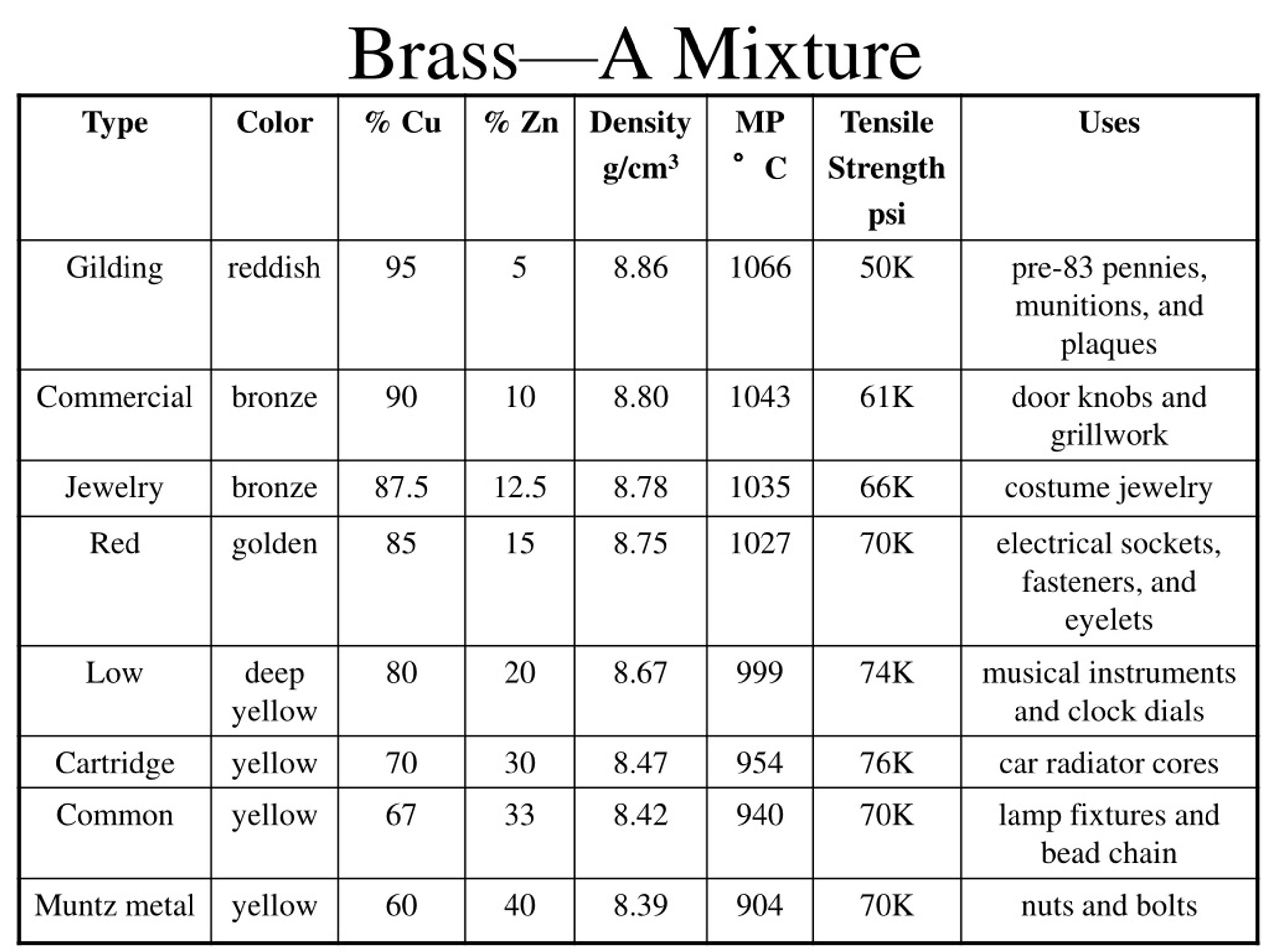
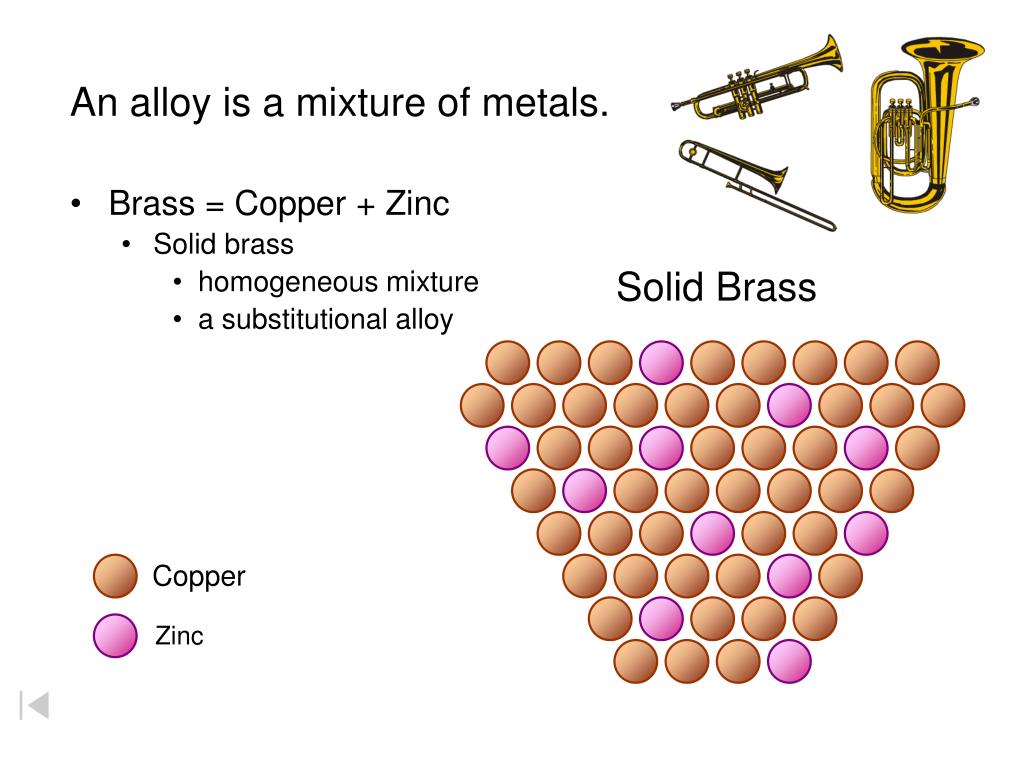
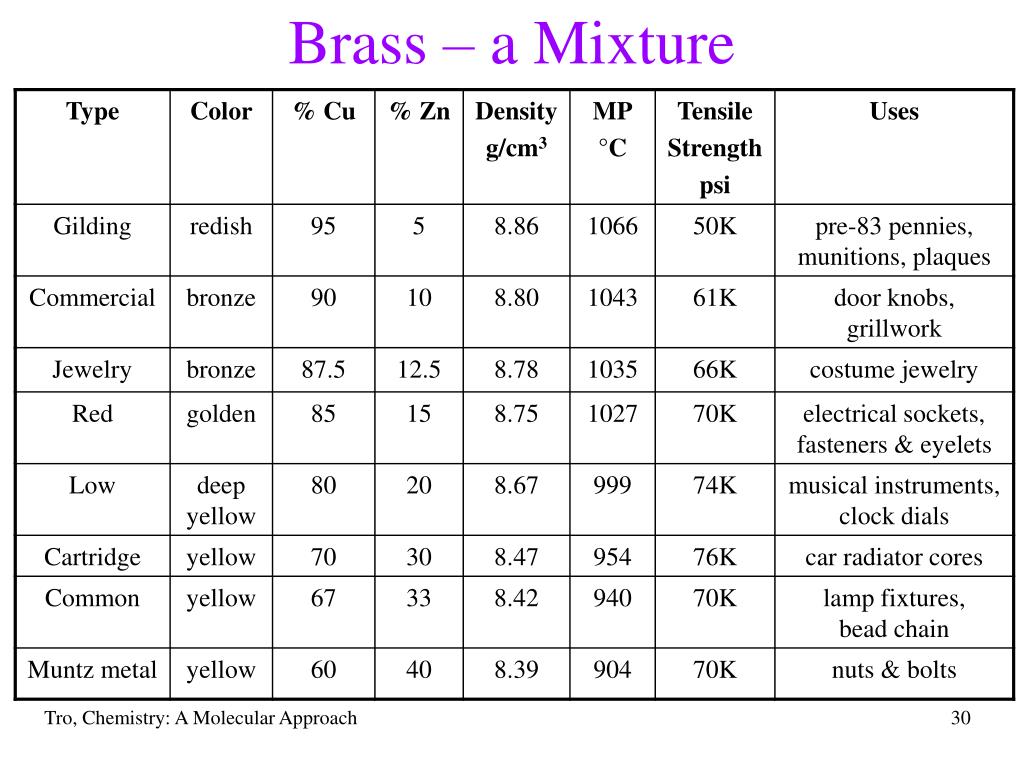
Alright, back to our golden-hued friend, brass. Brass is primarily made of two metals: copper and zinc. Copper brings its reddish-brown warmth, and zinc adds a touch of silvery-blue and helps make it stronger.
When brass is made, you melt down copper and zinc, stir 'em up real good, and then let 'em cool. They blend together, really, really well. Their atoms are dispersed evenly throughout the solid material. But here's the kicker: they haven't undergone a chemical reaction to form a completely new molecule, like hydrogen and oxygen did to make water.

Instead, they're like those really good roommates who just fit perfectly together. They occupy the same space, they're incredibly uniform, but if you zoomed in with super-duper microscopic vision, you'd still find copper atoms and zinc atoms, side-by-side, sharing electrons but not fundamentally changing each other's core identity.
This type of blend, especially when it involves metals, has a fancy name: an alloy. And an alloy is a type of... you guessed it! A mixture. Specifically, brass is often considered a solid solution, which is just a super uniform type of mixture where one solid is dissolved into another solid, much like sugar dissolves in water (but solidified!).

Why Does It Matter?
So, why all this talk about cakes and roommates? Because understanding that brass is a mixture (an alloy!) helps us appreciate why it behaves the way it does. Because it’s a mixture of copper and zinc, brass gets the best bits of both. It’s stronger and more durable than pure copper, and it’s more resistant to corrosion than pure zinc. It's also super malleable, making it perfect for bending into trumpets or etching into cool decorative pieces.
Think of it as the ultimate team-up. Copper and zinc, sharing their strengths without losing themselves. Just like a perfectly balanced trail mix that gives you energy and a sweet treat, brass combines its components to be incredibly useful and beautiful.
So next time you see something brass, give it a nod. It’s not a compound, doing all that intense chemical bonding. It’s just a really well-made, solid mixture – the cool, harmonious roommate situation of the metal world. And that, my friend, is something to smile about.