Unmasking Brass: Is It a Chemical Love Story or a Friendly Hangout?
Have you ever looked at a shiny trombone, a stately door knocker, or a gleaming antique and thought, "Wow, brass is just so... brassy!"?
But have you ever stopped to ponder what makes brass, well, brass? Is it a mysterious chemical concoction, a true compound, where everything has merged into a brand-new entity?
Or is it more like a harmonious group project, a delightful mixture, where different personalities come together but still retain their unique sparkle?
When Ingredients Become Best Friends Forever: The Compound Story
Let's talk about compounds for a second. Imagine throwing a magnificent party where all the guests arrive, and then BAM! They don't just mingle; they utterly transform.
They bond so tightly, so intimately, that they create something entirely new, with properties wildly different from their original selves. Think about water, H₂O, the ultimate compound celebrity!
You start with two gases, hydrogen (super flammable!) and oxygen (loves to make things burn!). They meet, they bond, and suddenly you have water, which puts out fires! That's a serious glow-up!
This isn't just a physical get-together; it's a deep, chemical embrace. The original ingredients literally lose their individual identities to form a unified, indivisible new substance.
It's like baking a cake where the flour, eggs, and sugar don't just sit next to each other; they chemically transform into a delicious, cohesive new dessert.
You can't just pick out the "egg" or the "flour" from a finished cake, can you? It's become one glorious, new thing!

When Everyone Retains Their Cool: The Mixture Marvel
Now, let's peek at the fantastic world of mixtures. This is a much more relaxed affair. Think of it like the most awesome potluck ever.
Everyone brings their incredible dish, they all sit together on the table, and they definitely enhance the dining experience, but each dish remains delightfully itself.
You can still identify the potato salad, the green bean casserole, and that suspiciously delicious Jell-O mold.
A classic example? A vibrant fruit salad! You've got strawberries, blueberries, kiwi, maybe some grapes. They're all together, looking fabulous, but each fruit is still undeniably a strawberry or a kiwi.
They haven't chemically bonded or morphed into a super-fruit. They're just physically hanging out, enjoying each other's company.
You could, with a little patience (and a fork!), even separate them back into their individual fruit piles if you really wanted to.

The individual components in a mixture retain their original properties. They haven't gone through a drastic chemical makeover; they're just sharing a space.
The Big Reveal: Is Brass a Compound or a Mixture? Drumroll Please!
Alright, moment of truth! Let's bring our shiny friend, brass, back into the spotlight.
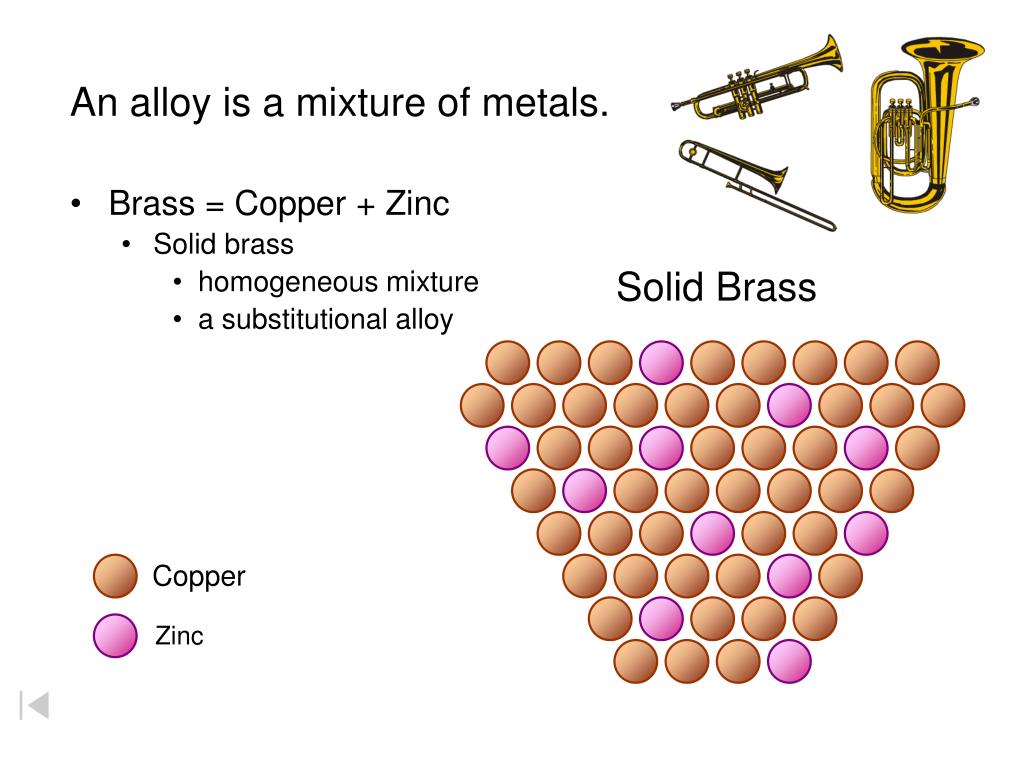
What is brass made of? It's primarily a dynamic duo of two different metals: lovely, reddish-gold copper and silvery-blue zinc.
When you melt these two metals down and mix them together, do they perform a chemical magic trick and become a brand-new, chemically distinct substance?
The answer, my delighted science enthusiast, is a resounding and enthusiastic: Brass is a Mixture! Specifically, it's a super cool type of mixture called an alloy.
Think of brass as the ultimate metallic smoothie! You've blended copper and zinc, but they haven't lost their fundamental "copper-ness" or "zinc-ness."
They haven't formed new chemical bonds like hydrogen and oxygen do in water. Instead, their atoms are just really, really well interspersed among each other.

Why Brass Is the Ultimate Mixture Maestro
One of the biggest clues that brass is a mixture is its variability. Just like you can make a fruit salad with more strawberries than blueberries, you can make different types of brass!
Some brass has a lot more copper and just a little zinc, making it redder. Other types might have nearly equal amounts, giving it a yellower hue.
This ability to vary the proportions of its ingredients without completely changing its fundamental identity is a hallmark of mixtures, not compounds.
If brass were a compound, its ingredients would have to be in fixed, precise ratios, like H₂O always having two hydrogen atoms for every one oxygen atom.
Also, if you had the right (and very intense!) scientific equipment, you could theoretically separate the copper and zinc back out of brass.
It wouldn't be easy – definitely not a kitchen science experiment! – but it's physically possible because they haven't formed unbreakable chemical bonds.

The copper still acts like copper, and the zinc still acts like zinc, even though they're intertwined. They've just created a new, enhanced material with properties that benefit from both.
The strength and workability of brass come from this close physical association, making it incredibly useful for everything from musical instruments to plumbing.
So, What Did We Learn From Our Brass Adventure?
Next time you see something made of brass, you can give it a knowing nod. You'll understand it's not a singular, chemically fused entity.
It's a fantastic example of teamwork, a brilliant partnership where copper and zinc decided to live together in metallic harmony.
They've created something stronger, more beautiful, and more versatile than either could be alone, without ever losing their individual metallic souls.
So let's raise a metaphorical glass (perhaps a brass one!) to the humble mixture, especially those dazzling alloys like brass, that bring so much function and sparkle to our world!
Science is truly everywhere, even in the most everyday objects, waiting for us to uncover its fun and fascinating secrets!